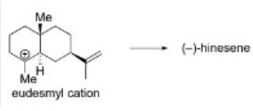
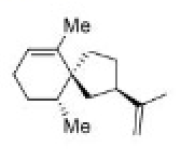
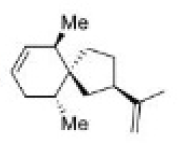
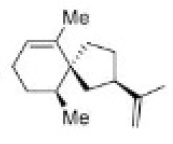
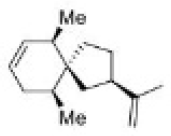
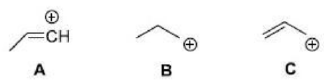
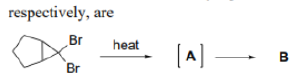
Correct option is C
A carbocation is any cation containing an even number of electrons in which a significant portion of the positive charge resides on a carbon atom.
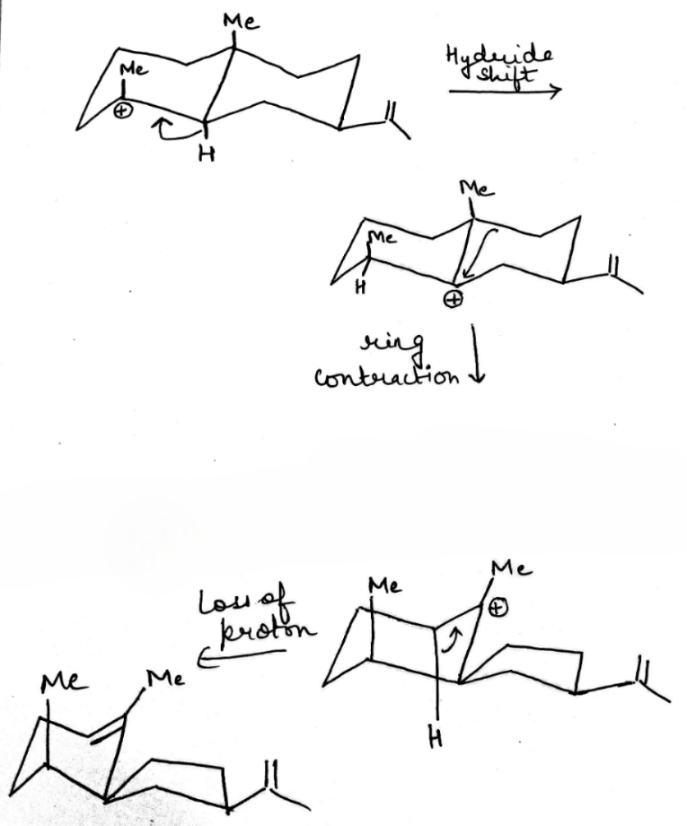
Carbocations can be stabilized by the migration of hydrogen, alkyl, or aryl groups, and occasionally, functional groups. Carbocations tend to change or shift their positive charge to the most stable configuration. This process is known as carbocation rearrangement.
In the first step, hydride migration occurs rather than hydrogen (or proton) because the H atom migrates with its pair of electrons. Thereafter, ring contraction converts the six membered ring to a five membered ring. Finally, loss of proton gives the desired product.