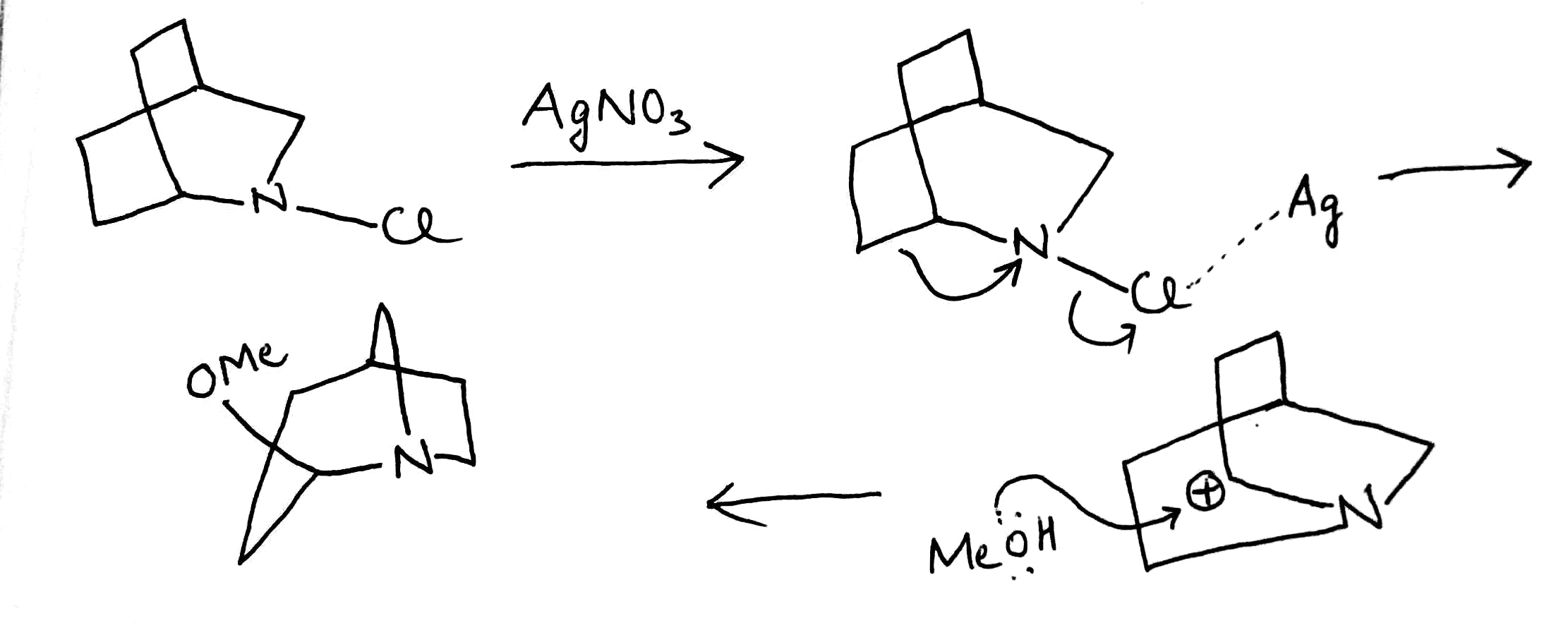
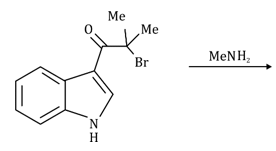
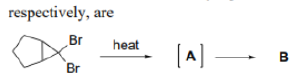
Correct option is B
In organic chemistry, neighbouring group participation (NGP, also known as anchimeric assistance) has been defined by the International Union of Pure and Applied Chemistry (IUPAC) as the interaction of a reaction centre with a lone pair of electrons in an atom or the electrons present in a sigma or pi bond contained within the parent molecule but not conjugated with the reaction centre. When NGP is in operation it is normal for the reaction rate to be increased.
NGP by aliphatic C-C or C-H bonds
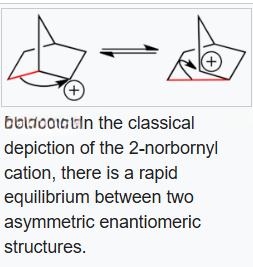
Aliphatic C-C or C-H bonds can lead to charge delocalization if these bonds are close and antiperiplanar to the leaving group. Corresponding intermediates are referred to a nonclassical ions, with the 2-norbornyl system as the most well known case.
A nonclassical ion usually refers to carbonium ions, a family of organic cations. They are characterized by delocalized three-center, two-electron bonds. The more stable members are often bi- or polycyclic. The term 2-norbornyl cation describes a carbonium ionic derivative of norbornane.
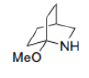
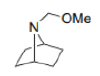
In the given reaction, Ag+ assists Cl-'s departure. As the nitrogen atom cannot bear a positive charge due to its high electronegativity, the neighbouring C-C bond simultaneously attacks the nitrogen atom.