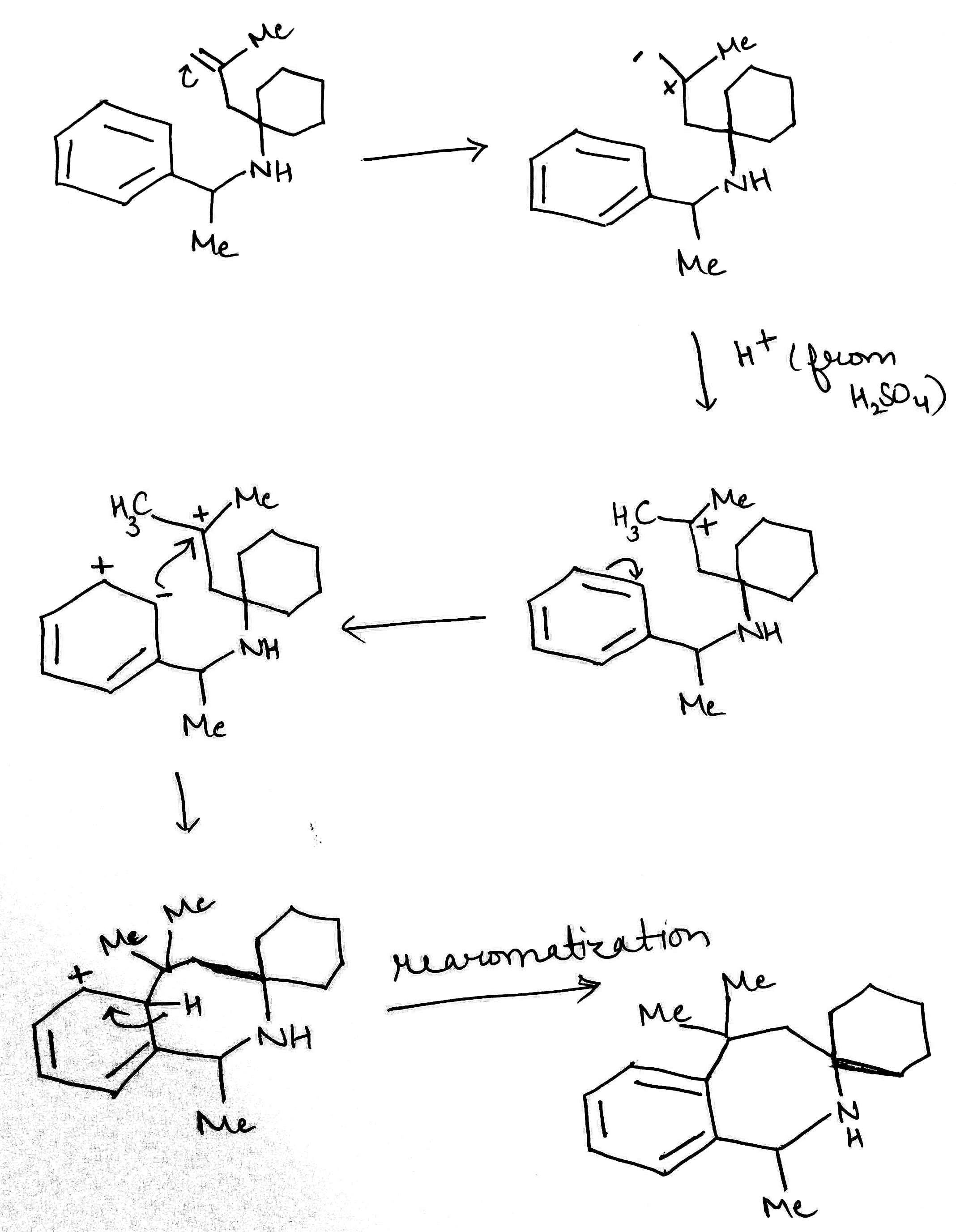
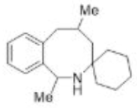
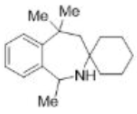
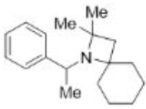
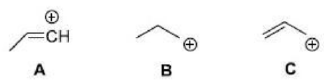
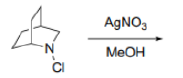
Correct option is B
The given reaction will proceed via the following steps:
1. Acid will protonate the alkene forming a more stable carbocation.
Stability of carbocations:
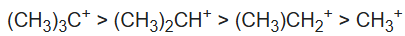
The three C–H σ bonds of the methyl group(s) attached to the carbocation can undergo the stabilization interaction but only one of them can be aligned perfectly with the empty p-orbital, depending on the conformation of the carbon–carbon bond. Donation from the two misaligned C–H bonds is weaker. The more adjacent methyl groups there are, the larger hyperconjugation stabilization is because of the increased number of adjacent C–H bonds.
2. An alkene bond of the benzene will attack the carbocation.
3. The benzene ring will undergo rearomatization.