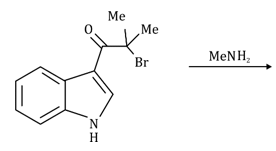
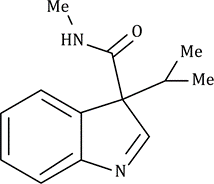
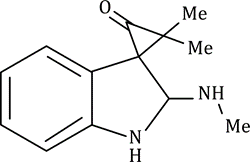
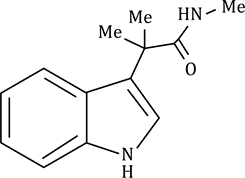
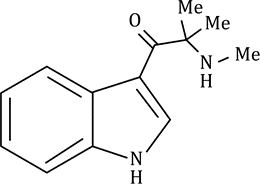
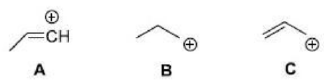
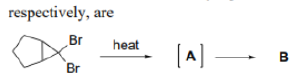
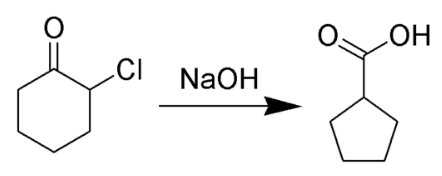
Correct option is C
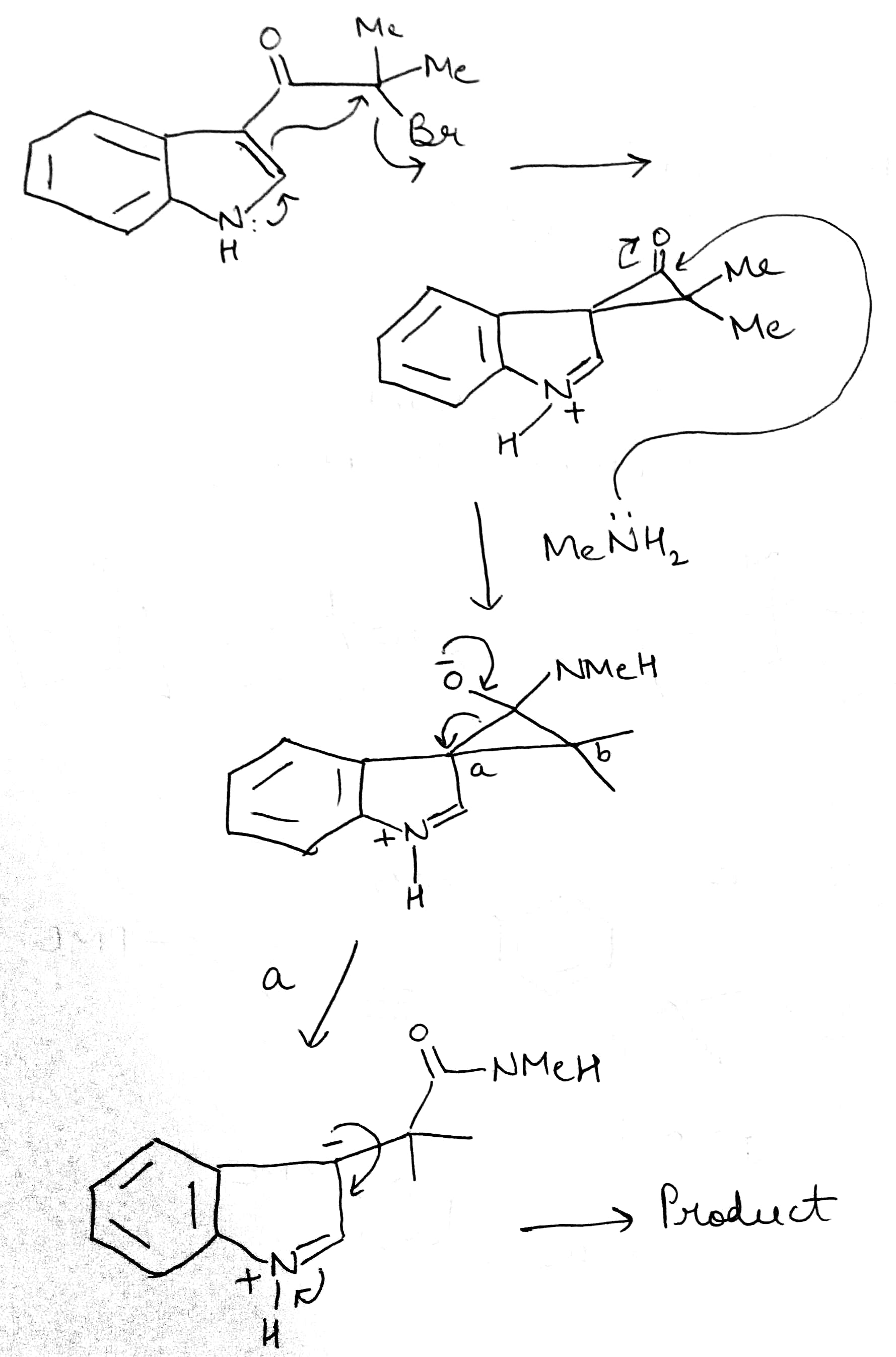
The Favorskii rearrangement is principally a rearrangement of cyclopropanones and α-halo ketones that leads to carboxylic acid derivatives. In the case of cyclic α-halo ketones, the Favorskii rearrangement constitutes a ring contraction. This rearrangement takes place in the presence of a base, sometimes hydroxide, to yield a carboxylic acid, but usually either an alkoxide base or an amine to yield an ester or an amide, respectively. α,α'-dihaloketones eliminate HX under the reaction conditions to give α,β-unsaturated carbonyl compounds.
Reaction mechanism
The reaction mechanism is thought to involve the formation of an enolate on the side of the ketone away from the chlorine atom. This enolate cyclizes to a cyclopropanone intermediate which is then attacked by the hydroxide nucleophile. Its formation can otherwise be viewed as a 2-electron electrocyclization of a 1,3-dipole, which can be captured in Diels Alder reactions. The cyclopropanone intermediate is opened to yield the more stable carbanion, which is quickly protonated.
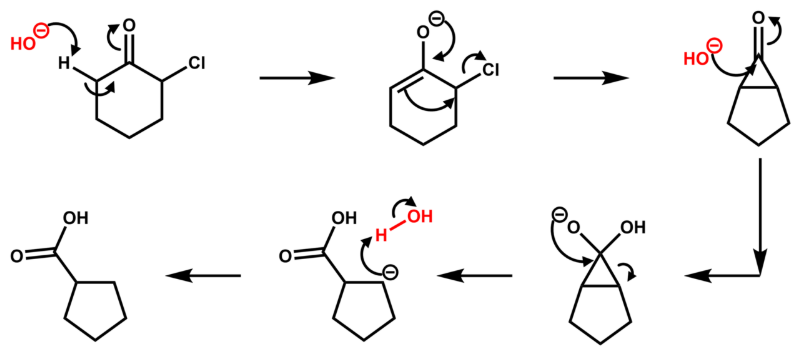
The second step has also been proposed to be stepwise process, with chloride anion leaving first to produce a zwitterionic oxyallyl cation before a disrotatory electrocyclic ring closure takes place to afford the cyclopropanone intermediate.