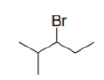
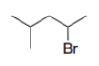
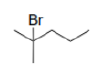
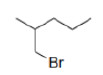
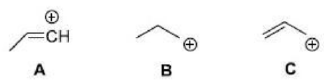
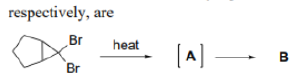
Correct option is C
There are a number of compounds whose homolysis is particularly important to chemists. They all have weak σ bonds, and generate radicals that can be put to some chemical use. The halogens are quite readily homolysed by light. These process are important in radical halogenation reactions.

Bromine can halogenate alkanes.
In this case, the first step of the radical chain reaction, the abstraction of H by Br•.
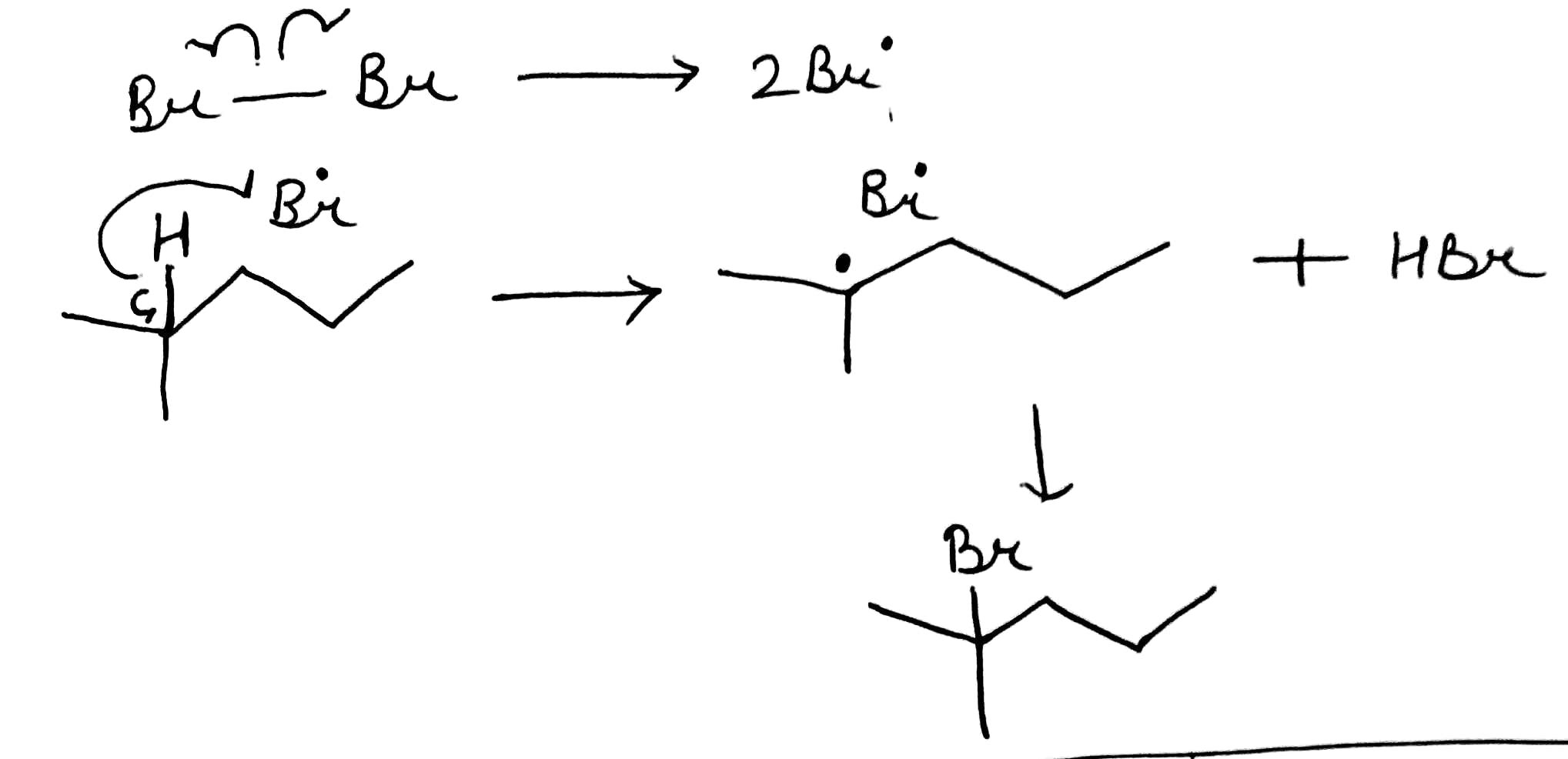
Tertiary radicals are relatively stable due to hyperconjugation. Hyperconjugation (σ-conjugation or no-bond resonance) refers to the delocalization of electrons with the participation of bonds of primarily σ-character. Usually, hyperconjugation involves the interaction of the electrons in a sigma (σ) orbital (e.g. C–H or C–C) with an adjacent unpopulated non-bonding p or antibonding σ* or π* orbitals to give a pair of extended molecular orbitals.