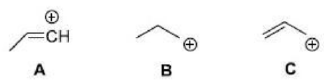
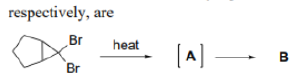
Correct option is B
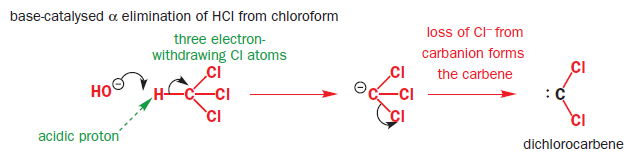
α Eliminations (eliminations in which both the proton and the leaving group are located on the same atom) are possible. In α Eliminations, a strong base removes an acidic proton adjacent to an electron withdrawing group to give a carbanion. Loss of a leaving group from the carbanion creates a carbene.
One of the best known α elimination reactions occurs when chloroform is treated with base. This is the most important way of making dichlorocarbene, :CCl2, and other dihalocarbenes too, although it must be said that the widespread use of dichlorocarbene in chemistry is due mainly to the ease with which it can be made using this method!
Carbenes can be divided into two types
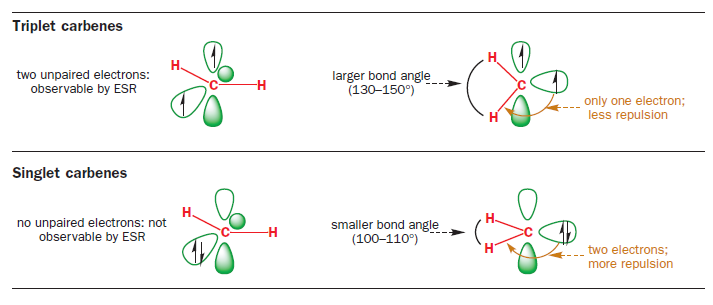
Spectroscopic investigations of a number of carbenes of differing structures have shown that they fall broadly into two groups: (1) those (which you will learn to call ‘triplets’) that ESR spectroscopy demonstrates have unpaired electrons and whose bond angles are 130–150°; and (2) those (like the stable crystalline carbene above which you will learn to call a ‘singlet’) that have bond angles of 100–110° but cannot be observed by ESR.
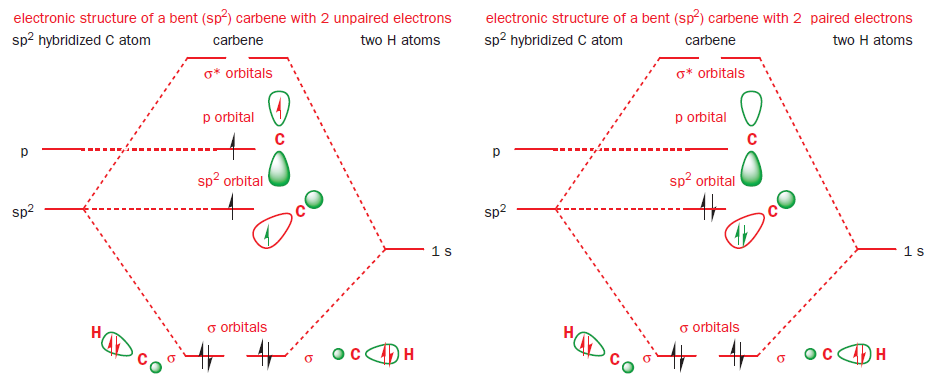
All these observations can be accounted for by considering the electronic structure of a carbene. Carbenes have 2-coordinate carbon atoms: you might therefore expect them to have a linear (diagonal) structure—like that of an alkyne—with an sp hybridized carbon atom.
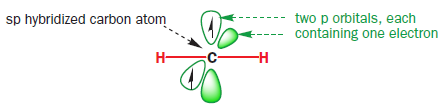
Such a linear carbene would have six electrons to distribute amongst two σ orbitals and two (higher-energy) p orbitals. The two electrons in the degenerate p orbitals would remain unpaired.
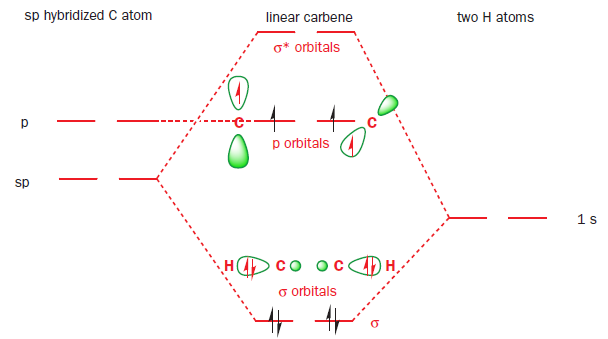
Yet few carbenes are linear: most are bent, with bond angles between 100° and 150°, suggesting a trigonal (sp2) hybridization state. An sp2 hybridized carbene would have three (lower-energy) sp2 orbitals and one (high-energy) p orbital in which to distribute its six electrons. There are two ways of doing this. Either all of the electrons can be paired, with each pair occupying one of the sp2 orbitals, or two of the electrons can remain unpaired, with one electron in each of the p orbitals and one of the sp2 orbitals.

These two possibilities explain our two observed classes of carbene, and the two possible arrangements of electrons (spin states) are termed triplet and singlet. The orbitals are the same in both cases but in triplet carbenes we have one electron in each of two molecular orbitals and in singlet carbenes both electrons go into the sp2 orbital.

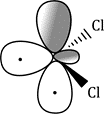
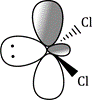
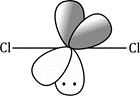
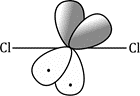
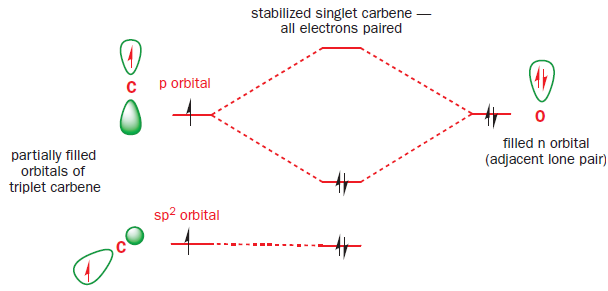
Carbenes that have singlet ground states (such as :CCl2) all have electron-rich substituents carrying lone pairs adjacent to the carbene centre. These lone pairs can interact with the p orbital of the carbene to produce a new, lower-energy orbital which the two electrons occupy. This stabilization of the lone pair provides the incentive that the electron in the p orbital needs to pair up in the sp2 orbital.
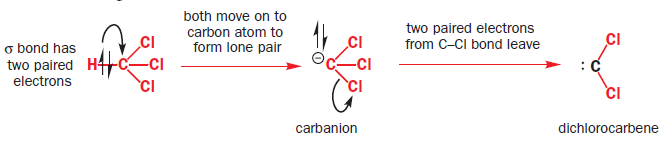
The starting material, a normal molecule of chloroform CHCl3, has all paired electrons. The C–H s bond breaks and the two paired electrons from it form the lone pair of the carbanion. The carbanion also has all paired electrons. The two paired electrons of one of the C–Cl bonds leaves the carbanion and the carbene is formed. It has two paired electrons in each of the two remaining C–Cl bonds and the lone pair, also paired. It is formed as a singlet.