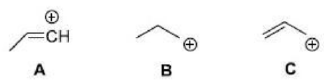
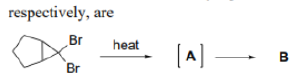
Correct option is B
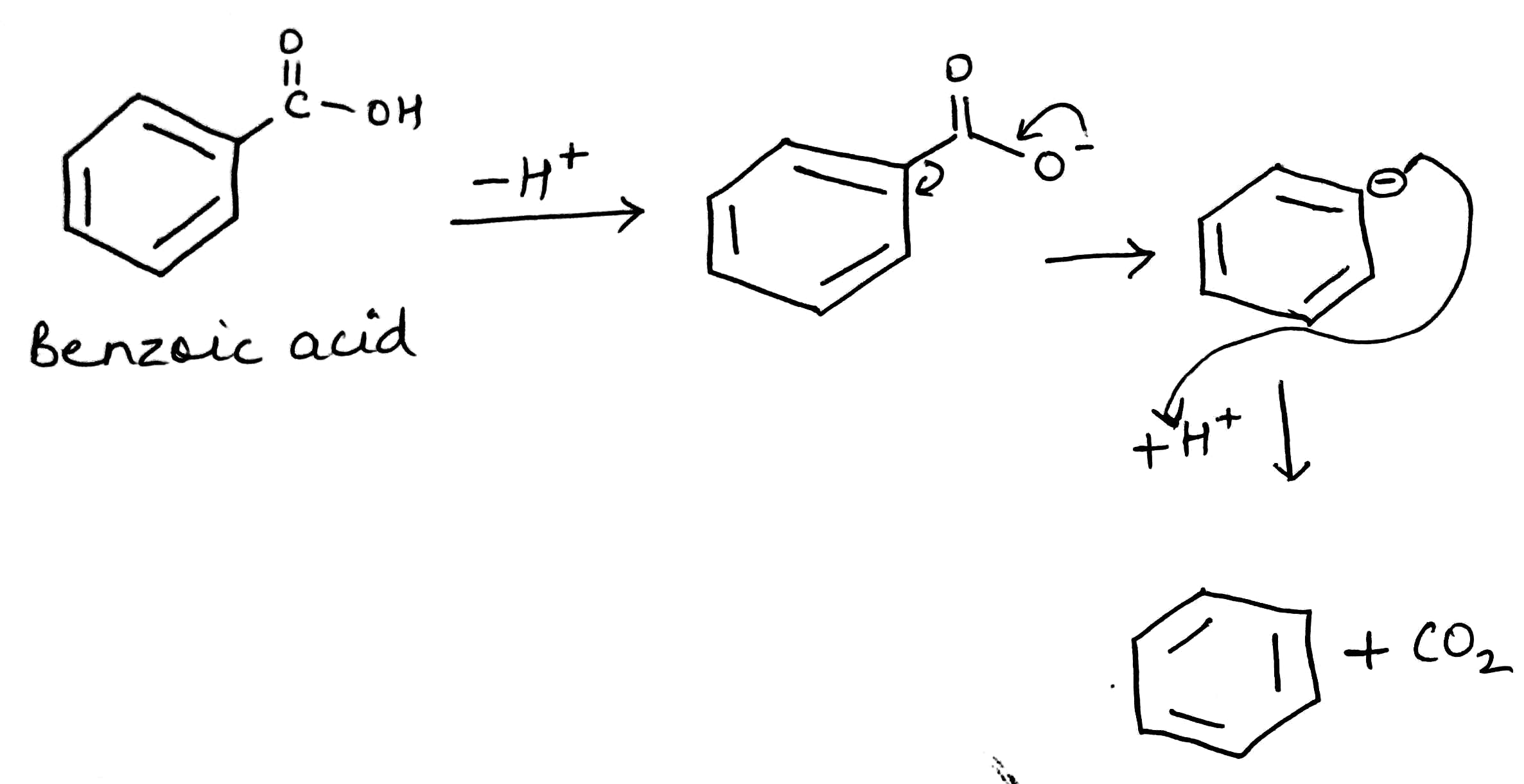
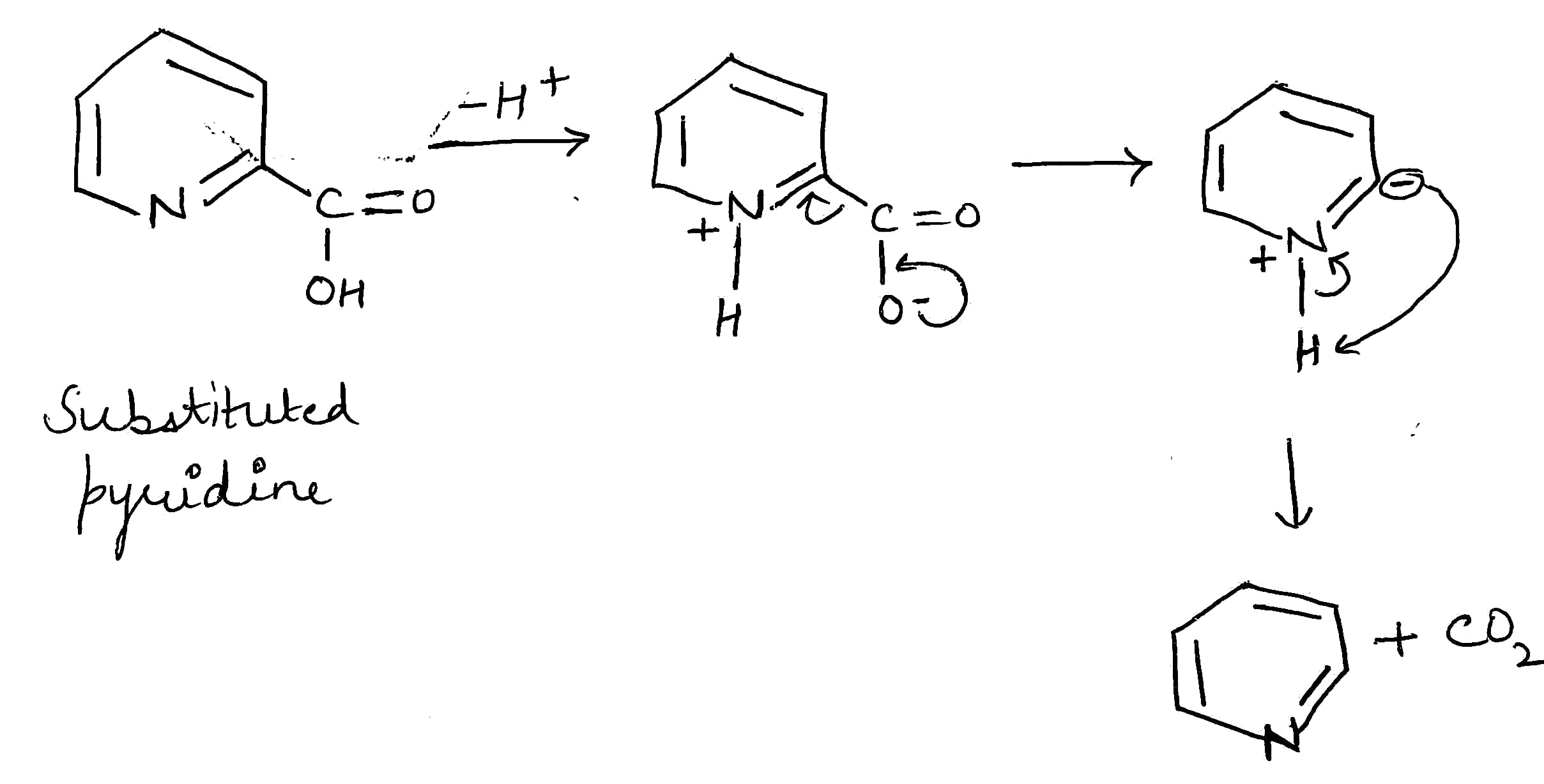
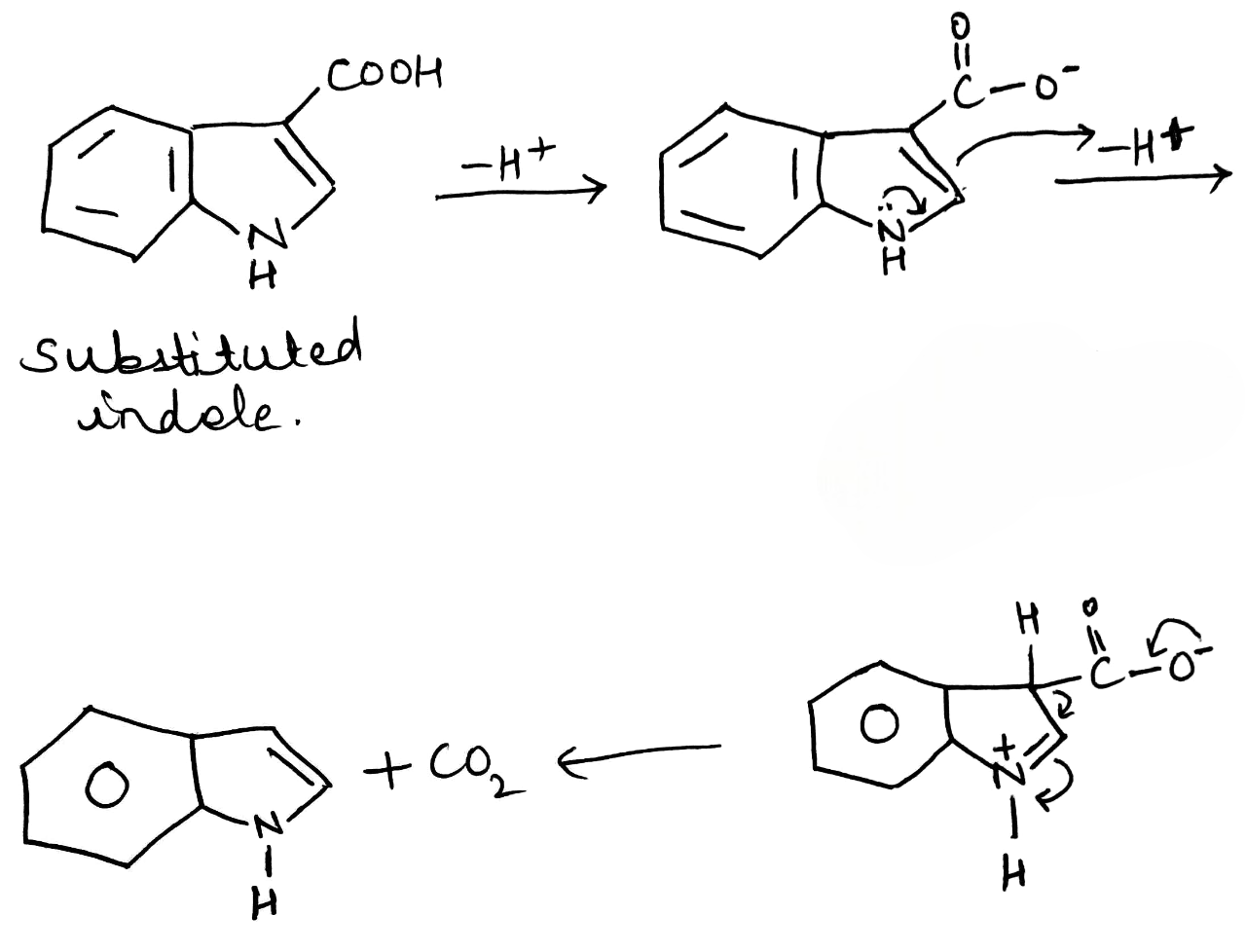
Decarboxylation is a chemical reaction that removes a carboxyl group and releases carbon dioxide (CO2). Usually, decarboxylation refers to a reaction of carboxylic acids, removing a carbon atom from a carbon chain.
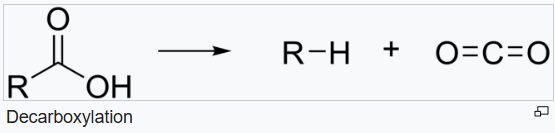
The rate of decarboxylation may be different for different molecules depending upon the nature of the substituent present in the molecule.
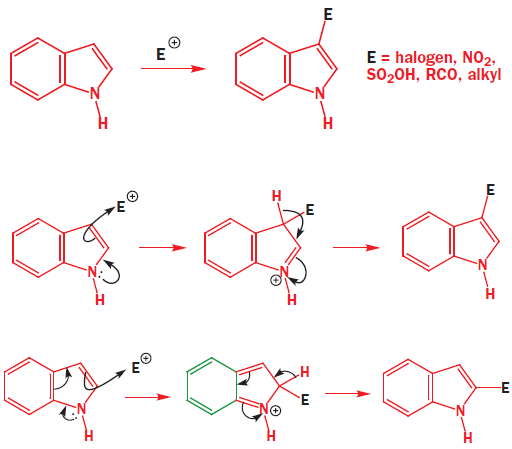
In indole, electrophilic substitution is preferred in the 3-position with almost all reagents. Halogenation, nitration, sulfonation, Friedel–Crafts acylation, and alkylation all occur cleanly at that position. A simple explanation is that reaction at the 3-position simply involves the rather isolated enamine system in the five-membered ring and does not disturb the aromaticity of the benzene ring. The positive charge in the intermediate is, of course, delocalized round the benzene ring, but it gets its main stabilization from the nitrogen atom. It is not possible to get reaction in the 2-position without seriously disturbing the aromaticity of the benzene ring.
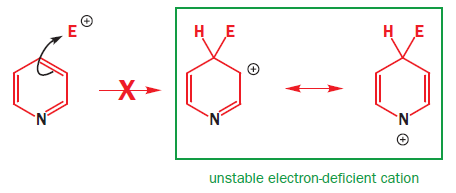
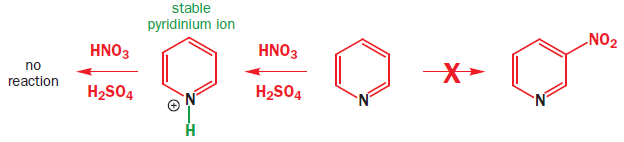
The lower energy of the orbitals of pyridine’s pi system means that electrophilic attack on the ring is difficult. Another way to look at this is to see that the nitrogen atom destabilizes the cationic would-be intermediate, especially at the 2- and 4-positions. An equally serious problem is that the nitrogen lone pair is basic and a reasonably good nucleophile-this is the basis for its role as a nucleophilic catalyst in acylations. The normal reagents for electrophilic substitution reactions, such as nitration, are acidic. Treatment of pyridine with the usual mixture of HNO3 and H2SO4 merely protonates the nitrogen atom. Pyridine itself is not very reactive towards electrophiles: the pyridinium ion is totally unreactive.