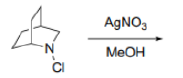
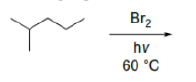
Correct option is D
Carbocation is a general term for ions with a positively charged carbon atom.
Factors affecting carbocation stability
Carbocations are at their most stable when the charge is on a tertiary carbon. At the same time, carbocations are the least stable when the charge is on a primary carbon. Carbocations tend to change or shift their positive charge to the most stable configuration. This process is known as carbocation rearrangement.
Resonance
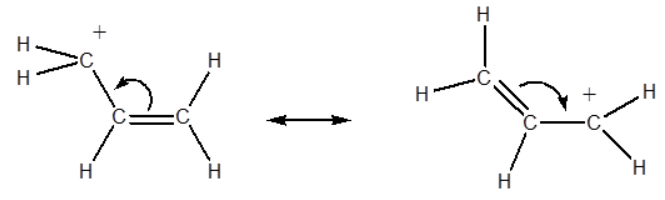
As the number of resonance structures increases, the stability of carbocations also increases. Furthermore, the greater the number of resonating structures, the greater the stability of the carbocations. The delocalization of the positive charge causes the electron loss to decrease further, thus increasing the overall stability.
Comparatively, it has been found that the resonance effect is a far greater influencing factor than substitution. The structures that have resonance are more stabilized than others.
Electronegativity
The electronegativity of an atom changes depending on the hybridization of the orbital employed in bonding. Electrons in s orbitals are held more tightly than electrons in p orbitals. Hence, a bond to an atom that employs an spx hybrid orbital for bonding will be more heavily polarized to that atom when the hybrid orbital has more s character. That is, when electronegativities are compared for different hybridization schemes of a given element, the order χ(sp3) < χ(sp2) < χ(sp) holds.
Hyperconjugation
Hyperconjugation (σ-conjugation or no-bond resonance) refers to the delocalization of electrons with the participation of bonds of primarily σ-character. Usually, hyperconjugation involves the interaction of the electrons in a sigma (σ) orbital (e.g. C–H or C–C) with an adjacent unpopulated non-bonding p or antibonding σ* or π* orbitals to give a pair of extended molecular orbitals.
Stability of carbocations:
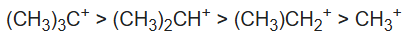
The three C–H σ bonds of the methyl group(s) attached to the carbocation can undergo the stabilization interaction but only one of them can be aligned perfectly with the empty p-orbital, depending on the conformation of the carbon–carbon bond. Donation from the two misaligned C–H bonds is weaker. The more adjacent methyl groups there are, the larger hyperconjugation stabilization is because of the increased number of adjacent C–H bonds.
The inductive effect in a molecule is a local change in the electron density due to electron-withdrawing or electron-donating groups elsewhere in the molecule, resulting in a permanent dipole in a bond. It is present in a σ (sigma) bond.
Alkyl groups tend to donate electrons, leading to the +I effect.
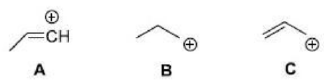
In carbocation A, the positive charge resides on a sp hybridized carbon atom.
In carbocation B, the positive charge resides on a sp2 hybridized carbon atom. This carbocation is stabilized by hyperconjugation and inductive effect of the ethyl group.
In carbocation C, the positive charge resides on a sp2 hybridized carbon atom. This carbocation undergoes resonance stabilization.