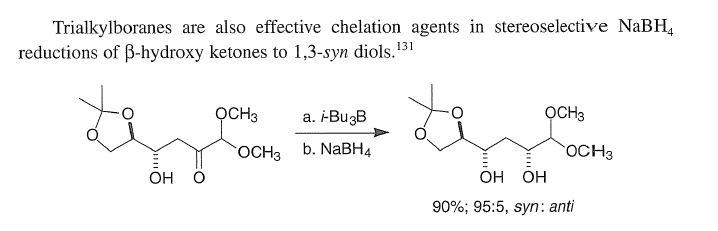
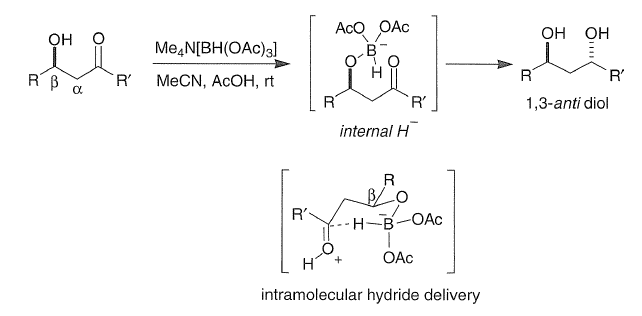
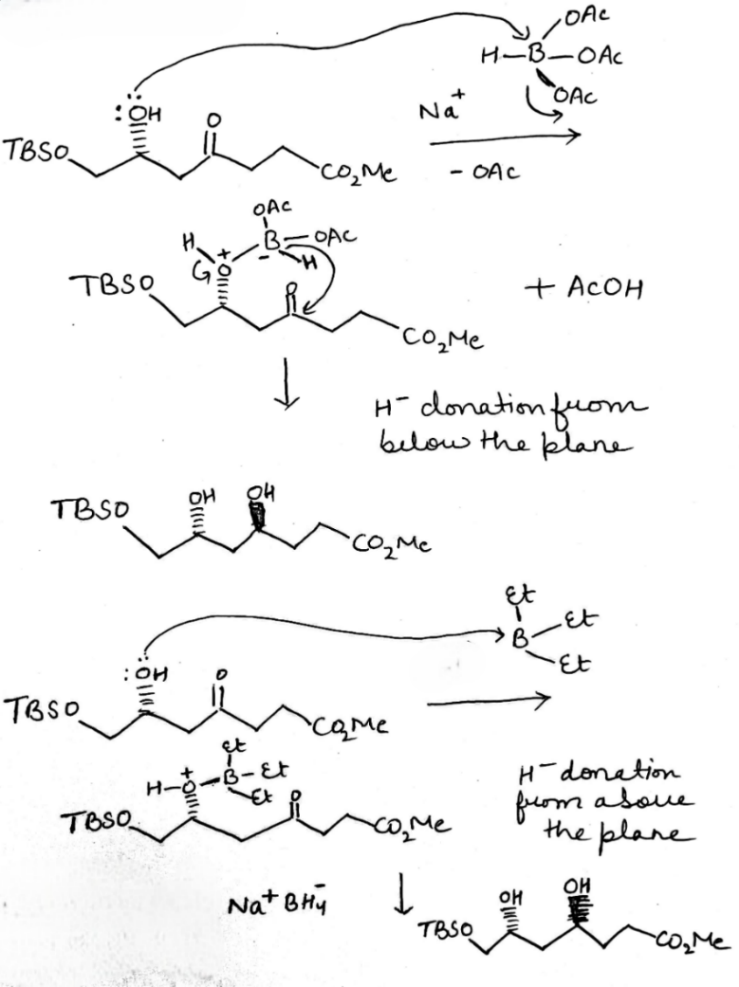
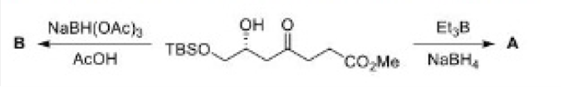
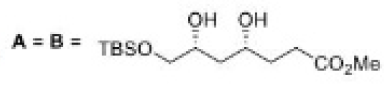
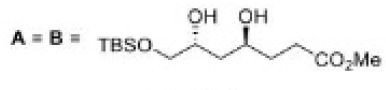
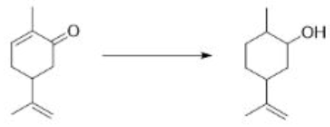
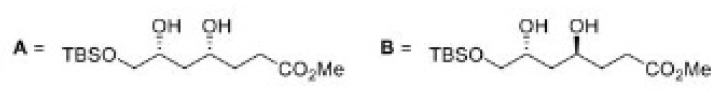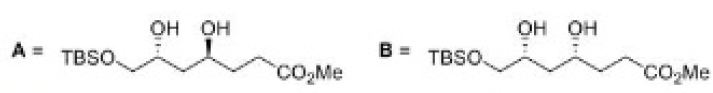Correct option is C
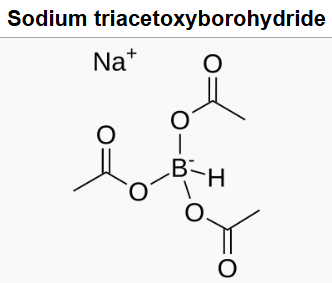
Sodium triacetoxyborohydride, also known as sodium triacetoxyhydroborate, commonly abbreviated STAB, is a chemical compound with the formula Na[(CH3COO)3BH].Like other borohydrides, it is used as a reducing agent in organic synthesis. Sodium triacetoxyborohydride is a milder reducing agent than sodium borohydride. It reduces aldehydes but not most ketones.
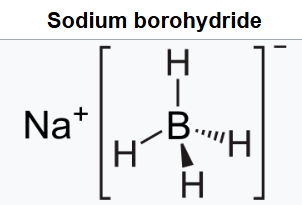
Sodium borohydride, also known as sodium tetrahydridoborate and sodium tetrahydroborate, is an inorganic compound with the formula NaBH4.NaBH4 reduces many organic carbonyls, depending on the conditions. Most typically, it is used in the laboratory for converting ketones and aldehydes to alcohols.
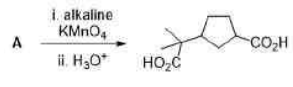
In the formation of A, due to the presence of bulky group below the plane, H- will attack from above the plane and therefore OH group goes below the plane.
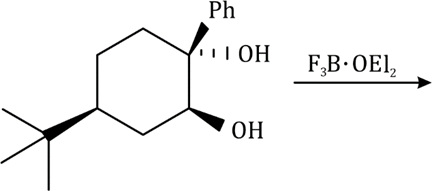
In the formation of B, due to the presence of bulky group (containing H-) below the plane, the attack of H- will occur from below the plane and therefore OH group goes above the plane.