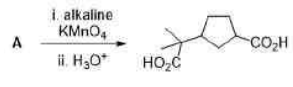
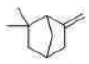
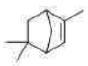
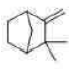
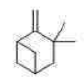
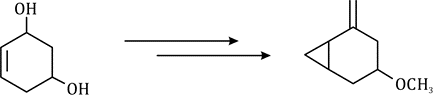
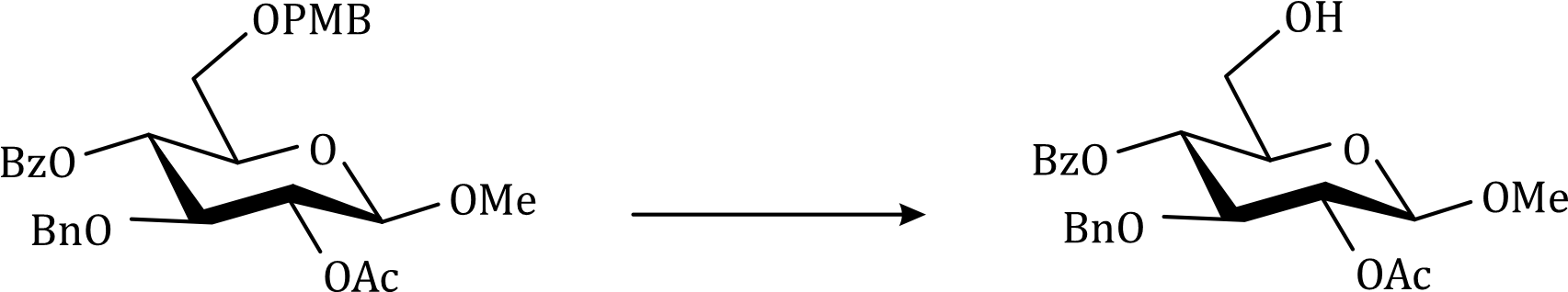
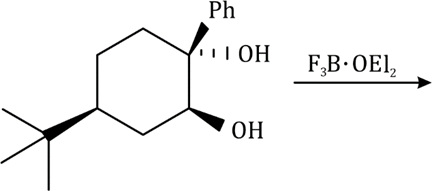
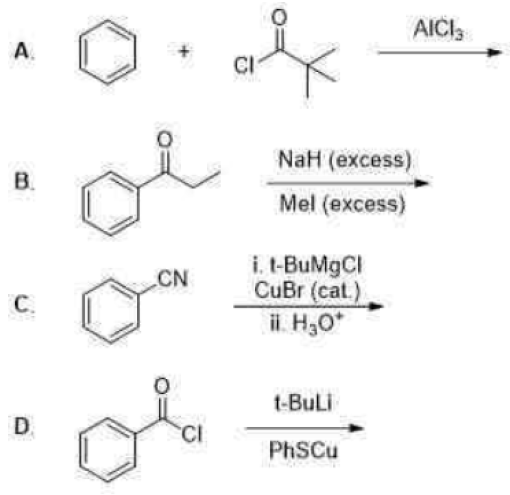
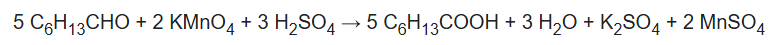Correct option is C
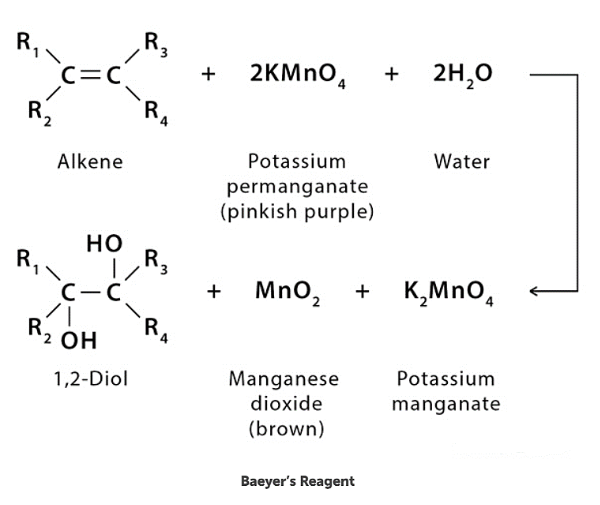
Baeyer’s reagent is a cold potassium permanganate alkaline solution (KMnO4). It is a strong oxidising agent that is utilized in qualitative organic analysis to check for unsaturation. When this solution reacts with an unsaturated compound (such as an alkene or alkyne), the colour changes from pinkish-purple to brown.
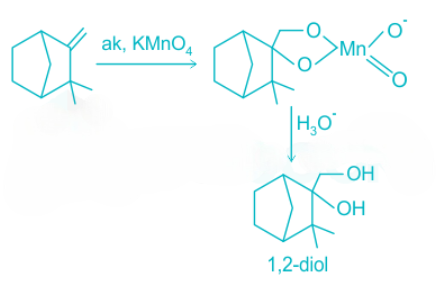
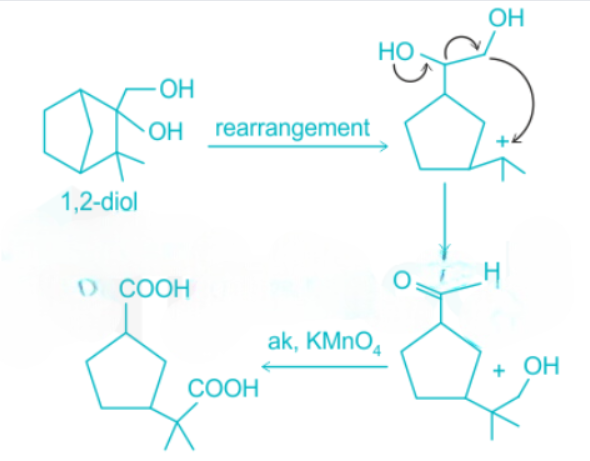
1,2-diol undergoes rearrangement and then is oxidized to carboxylic acid.
Potassium permanganate oxidizes aldehydes to carboxylic acids, illustrated by the conversion of n-heptanal to heptanoic acid: