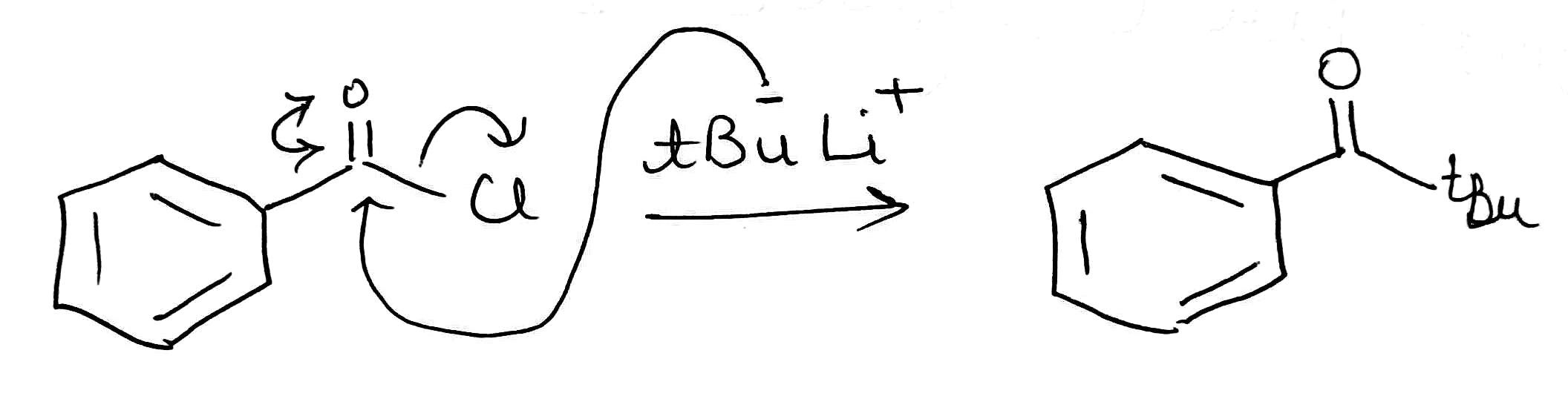
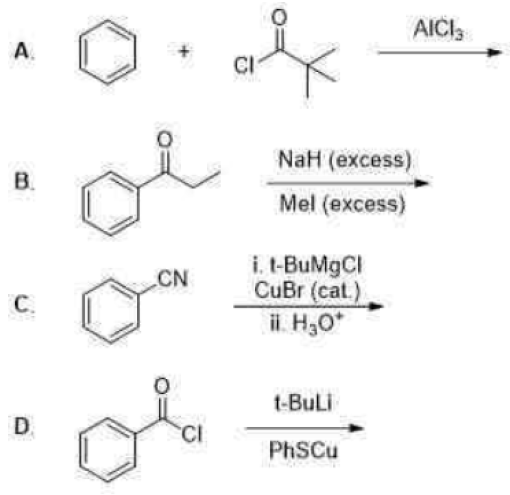
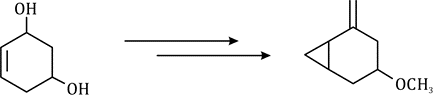
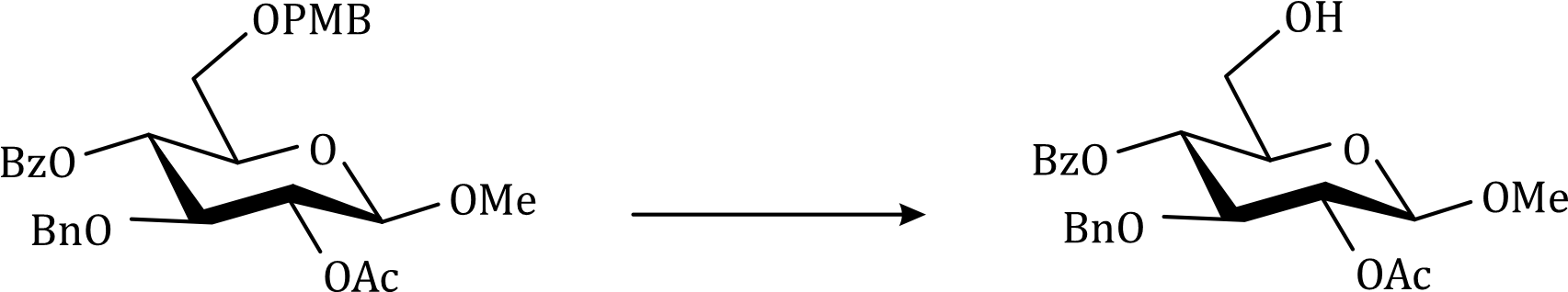
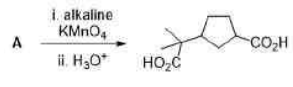
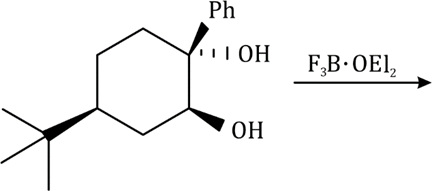
Correct option is B
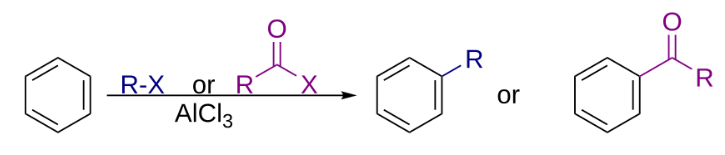
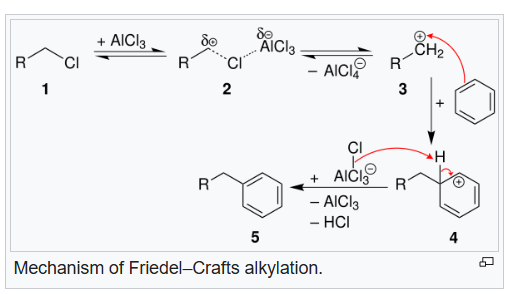
The Friedel–Crafts reactions are a set of reactions developed to attach substituents to an aromatic ring. Friedel–Crafts reactions are of two main types: alkylation reactions and acylation reactions. Both proceed by electrophilic aromatic substitution.
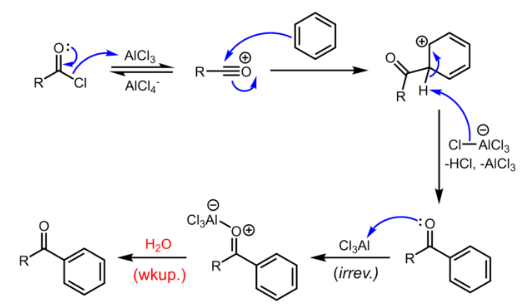
Mechanism of Friedel-Crafts acylation
The reaction proceeds through generation of an acylium center. The reaction is completed by deprotonation of the arenium ion by AlCl4-, regenerating the AlCl3 catalyst. However, in contrast to the truly catalytic alkylation reaction, the formed ketone is a moderate Lewis base, which forms a complex with the strong Lewis acid aluminum trichloride. The formation of this complex is typically irreversible under reaction conditions. Thus, a stochiometric quantity of AlCl3 is needed. The complex is destroyed upon aqueous workup to give the desired ketone.
A:
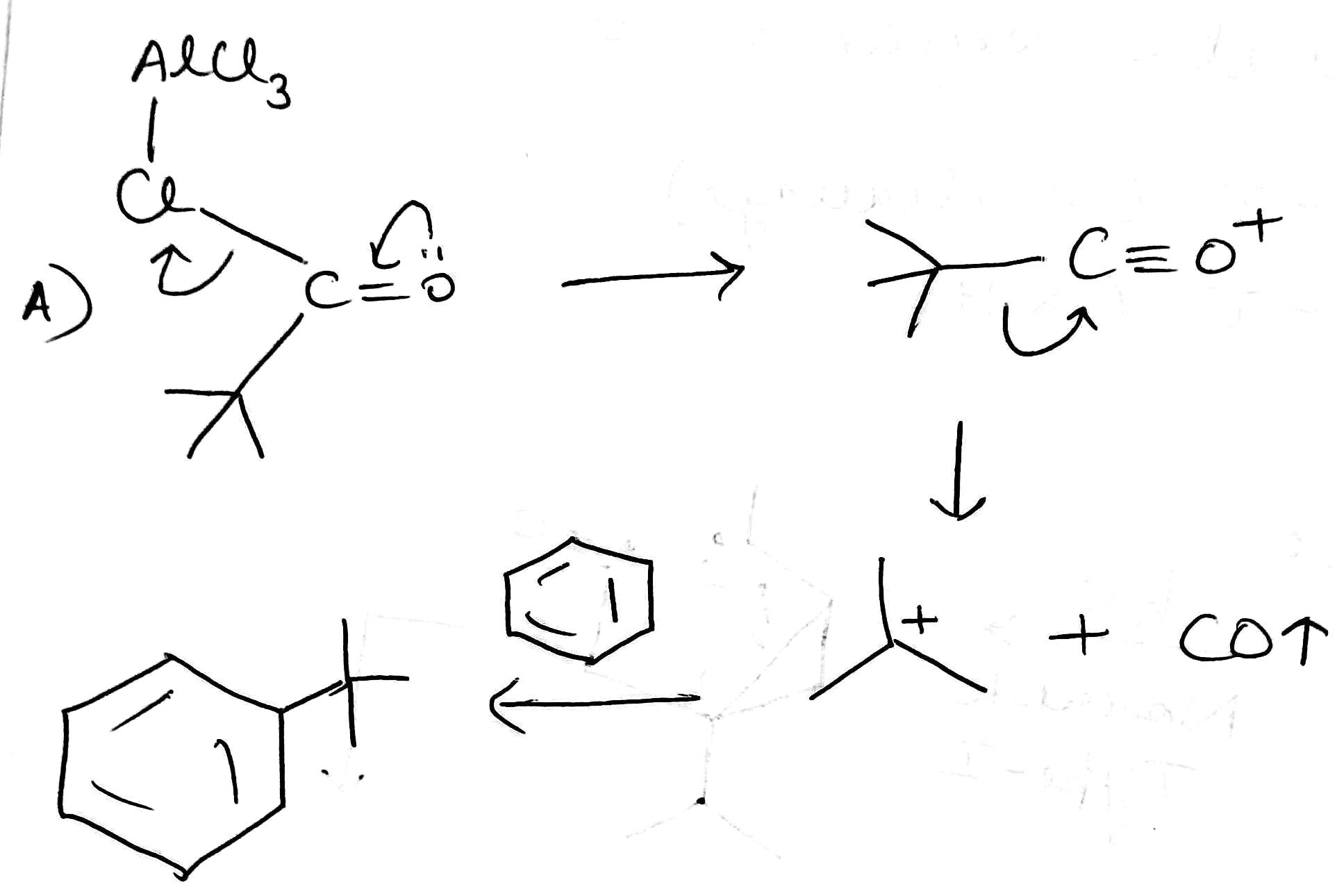
Stability of carbocations:
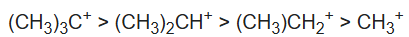
The three C–H σ bonds of the methyl group(s) attached to the carbocation can undergo the stabilization interaction but only one of them can be aligned perfectly with the empty p-orbital, depending on the conformation of the carbon–carbon bond. Donation from the two misaligned C–H bonds is weaker. The more adjacent methyl groups there are, the larger hyperconjugation stabilization is because of the increased number of adjacent C–H bonds.
B:Sodium hydride is the chemical compound with the empirical formula NaH. This alkali metal hydride is primarily used as a strong yet combustible base in organic synthesis. NaH notably deprotonates carbon acids (i.e., C-H bonds) such as 1,3-dicarbonyls such as malonic esters. The resulting sodium derivatives can be alkylated.
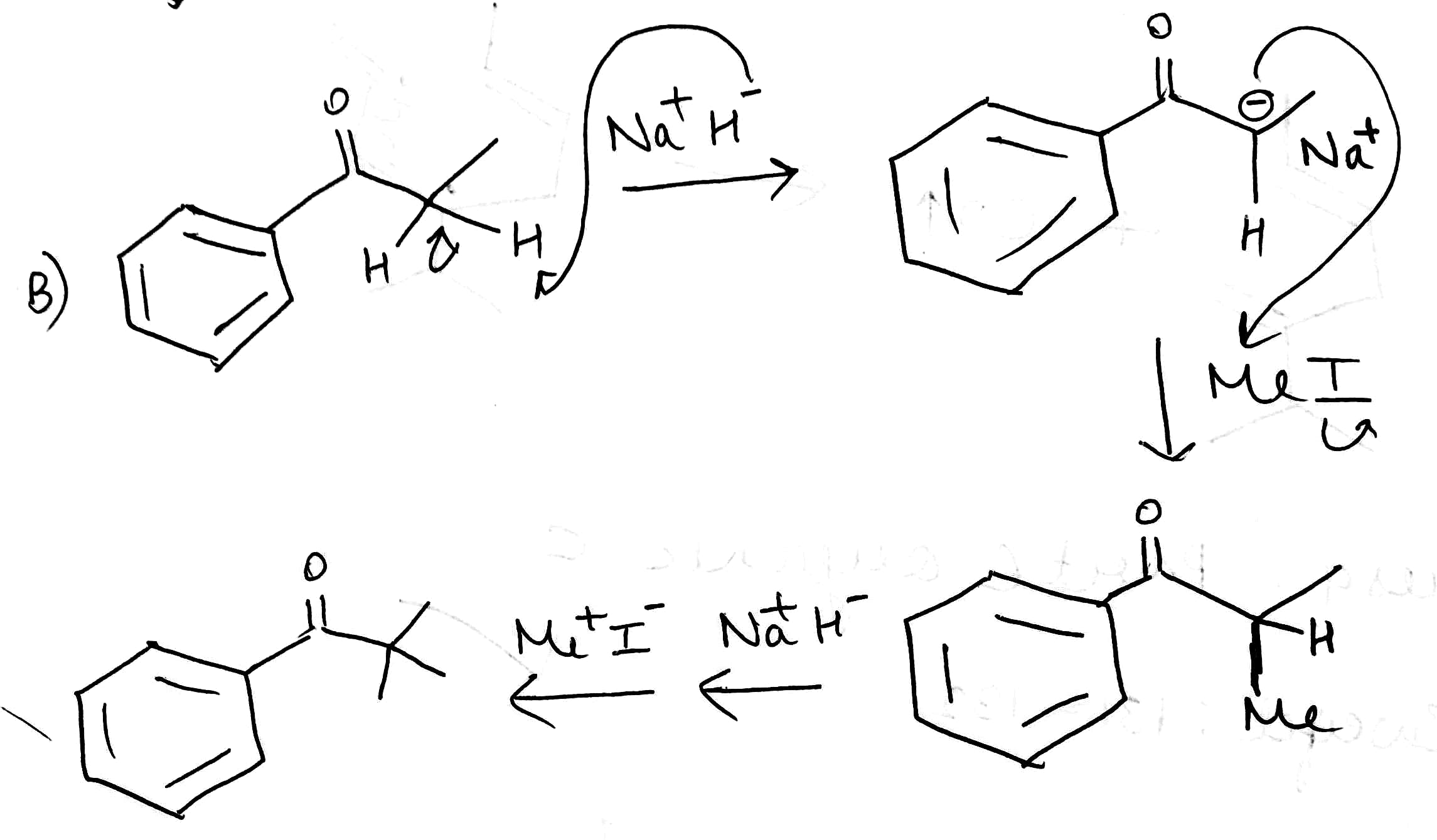
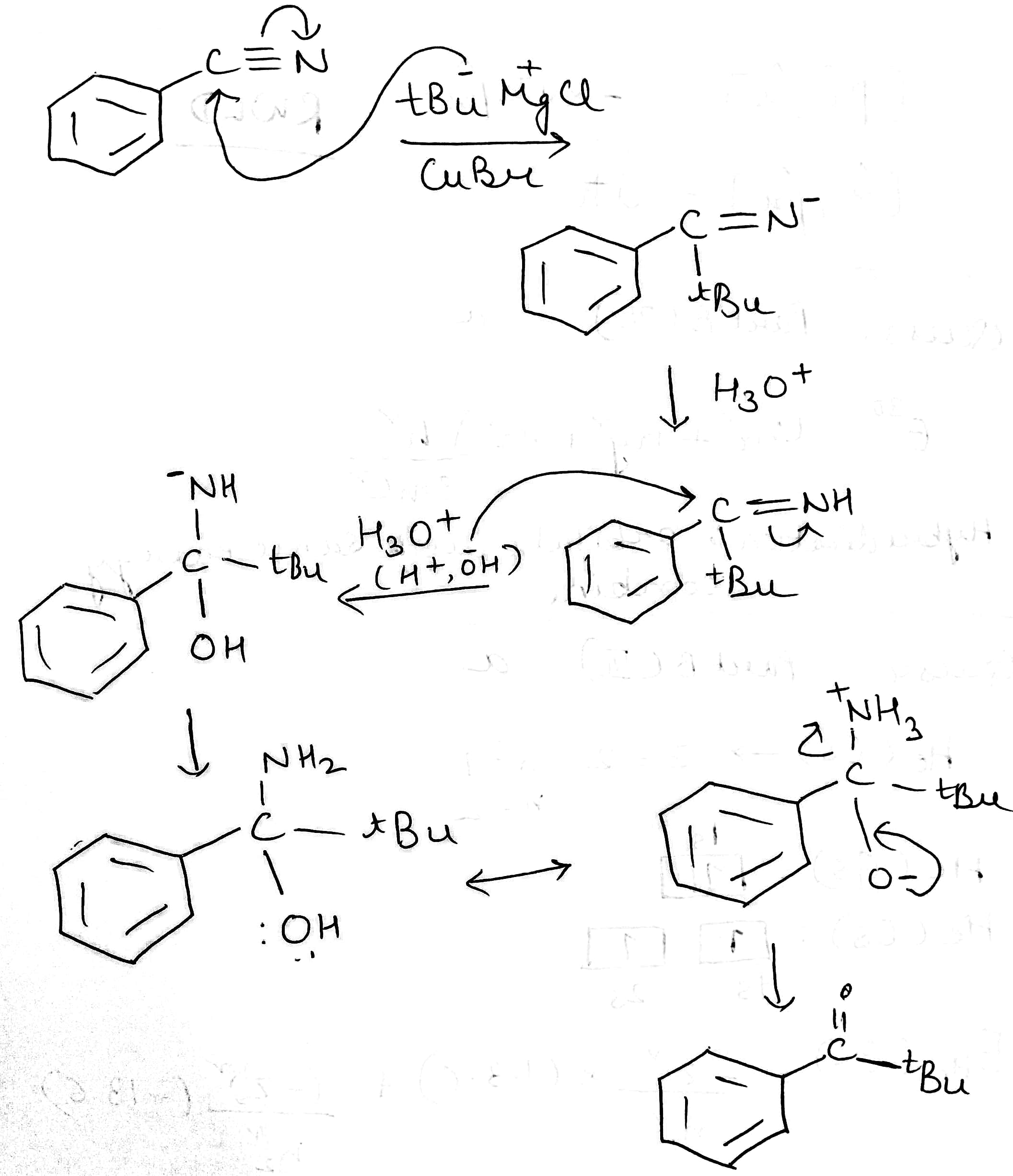
C. Benzonitrile reacts with t-butyl magnesium chloride in presence of CuBr to give t-BuCOPh.
D. The given reaction involves benzoyl chloride (C₆H₅COCl) as the starting material, treated with tert-butyllithium (t-BuLi).