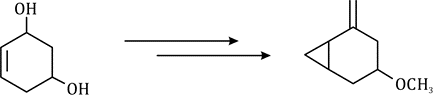
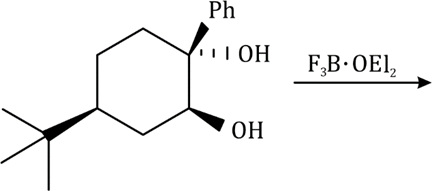
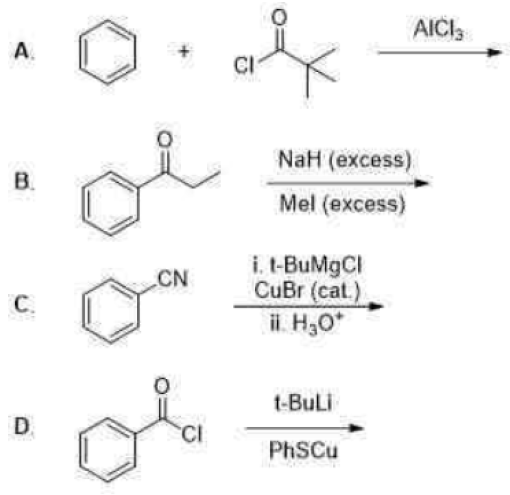
Correct option is A
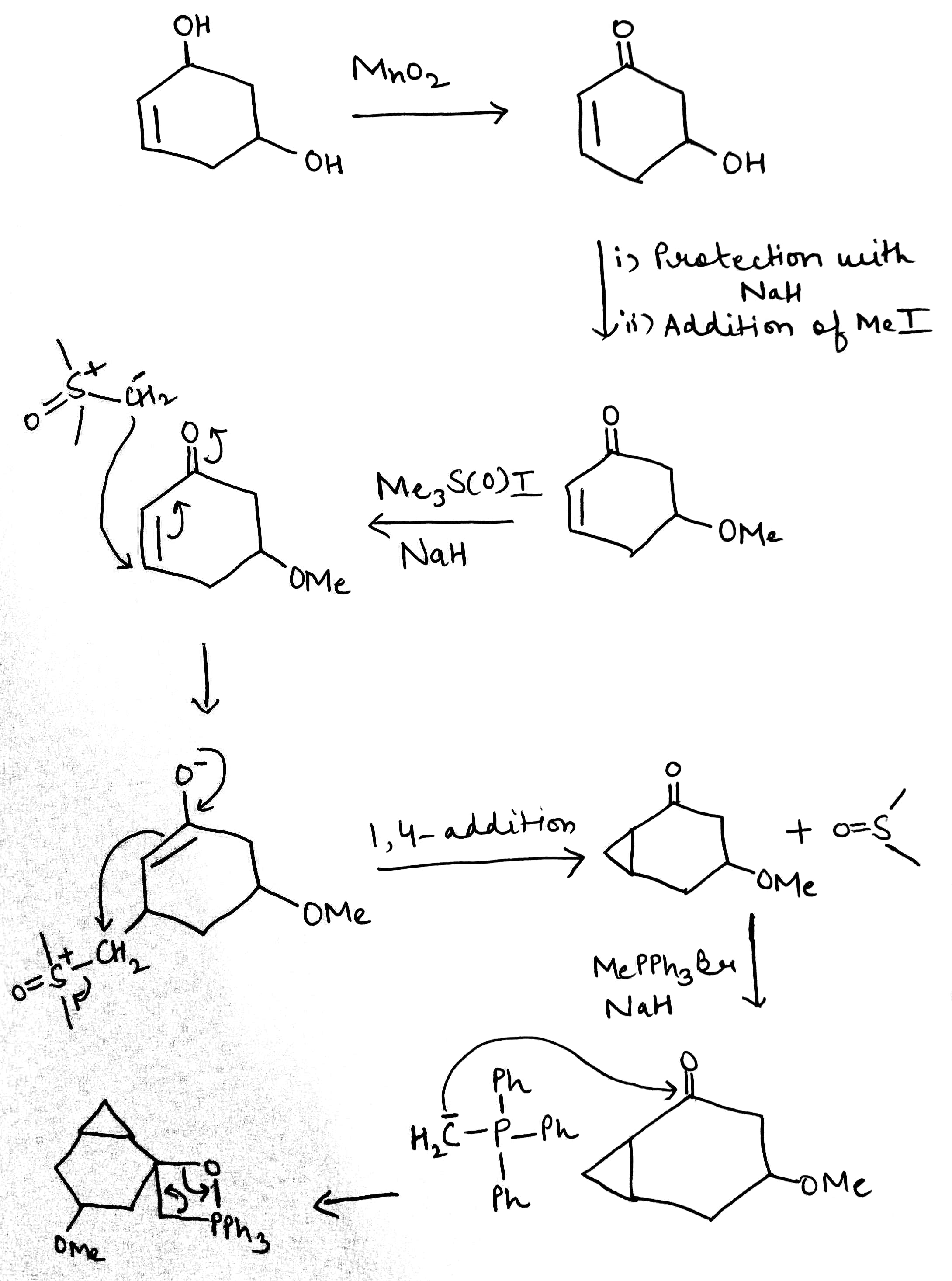
MnO2
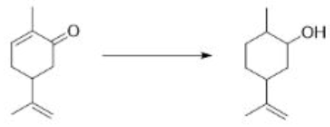
A useful, mild reagent for the oxidation of primary and secondary alcohols to carbonyl compounds is manganese dioxide. This reagent has found most use as a highly specific oxidant for allylic and benzylic hydroxy groups, and reaction takes place under mild conditions (room temperature) in a neutral solvent (e.g. water, petroleum, acetone, DMF, CH2Cl2 or CHCl3).
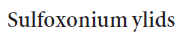
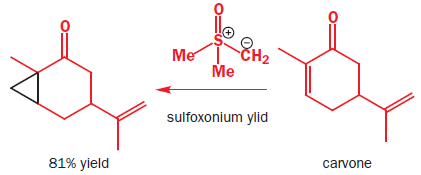
There is a very important class of stabilized sulfur ylids that owe their stability not to an additional anion-stabilizing substituent but to a more anion-stabilizing sulfur group. These are the sulfoxonium ylids, made from dimethylsulfoxide by SN2 substitution with an alkyl halide. Note that the sulfur atom is the nucleophile rather than the oxygen atom in spite of the charge distribution. The high-energy sulfur lone pair is better at SN2 substitution at saturated carbon—a reaction that depends very little on charge attraction.
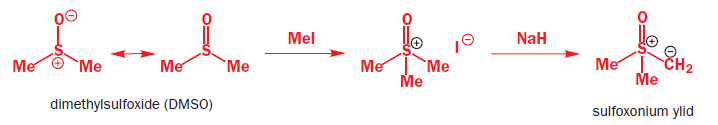
Sulfoxonium ylids react with unsaturated carbonyl compounds in the same way as the stabilized ylids—they form cyclopropanes rather than epoxides.
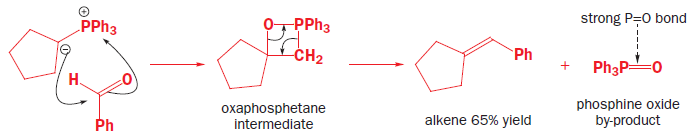
The Wittig reaction is like the base-promoted Peterson reaction: it is a syn elimination, driven by the strength of an oxygen–heteroatom bond, but in this case the heteroatom is phosphorus. Phosphorus atoms, especially those that are positively charged or that carry electronegative substituents, can increase the acidity of protons adjacent to them on the carbon skeleton. Phosphonium salts (made in a manner analogous to the formation of ammonium salts from amines, in other words, by reaction of an alkyl halide with a phosphine) can therefore be deprotonated by a moderately strong base to give a species known as an ylid, carrying (formally) a positive and a negative charge on adjacent atoms. Ylids can alternatively be represented as doubly bonded species, called phosphoranes.
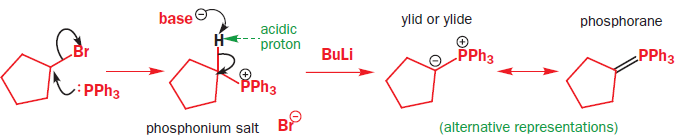
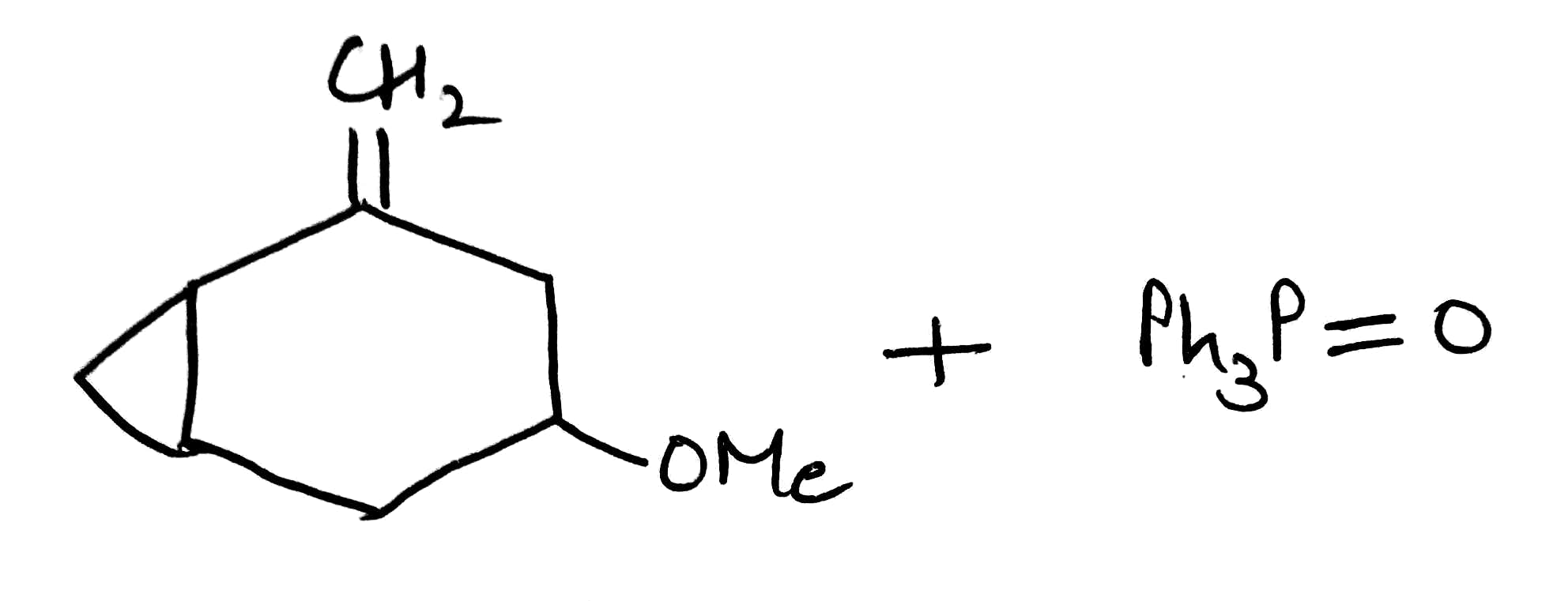
Ylids can be isolated, but are usually used in reactions immediately they are formed. They are nucleophilic species that will attack the carbonyl groups of aldehydes or ketones, generating the four-membered ring oxaphosphetane intermediates. Oxaphosphetanes are unstable: they undergo elimination to give an alkene (65% yield for this particular example) with a phosphine oxide as a byproduct. The phosphorus–oxygen double bond is extremely strong and it is this that drives the whole reaction forward.