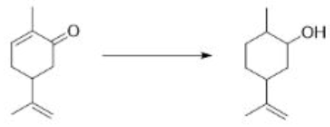
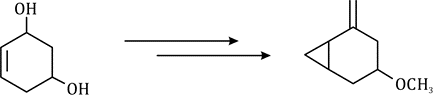
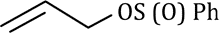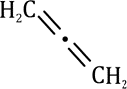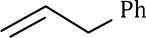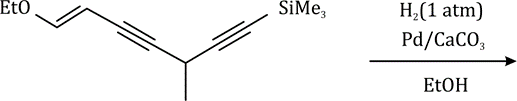Correct option is A
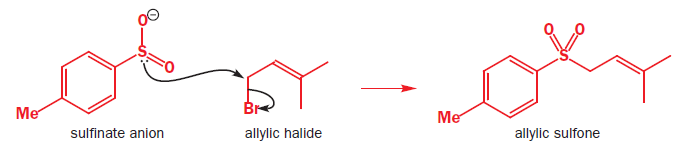
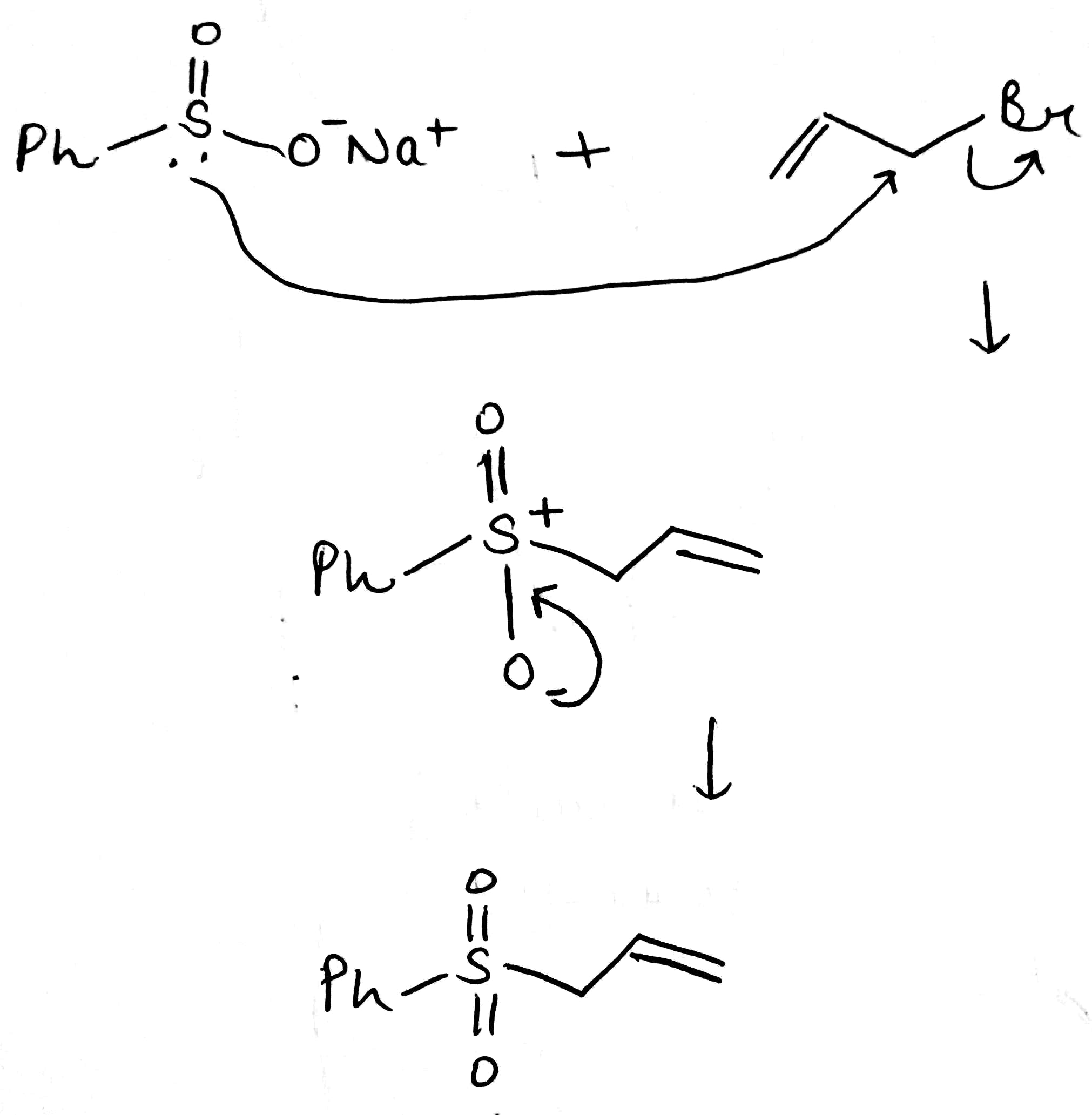
Sulfur at the S(II) oxidation state is both a good nucleophile and a good electrophile. This is also true at higher oxidation states though the compounds become harder electrophiles as the positive charge on sulfur increases.
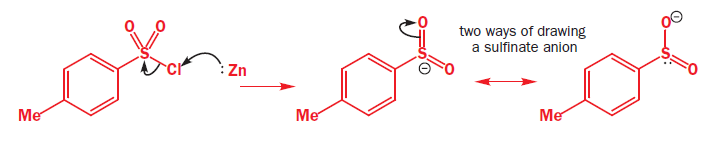
In the above reaction tosyl (toluene-para-sulfonyl) chloride [TsCl], reacts with Zn metal. Zinc provides two electrons and turns the compound into an anion. Surprisingly, this anion is also a good soft nucleophile and attacks saturated carbon atoms through the sulfur atom.