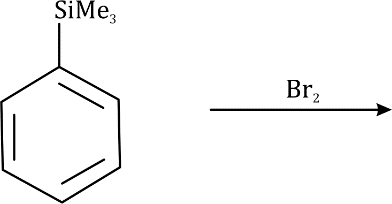
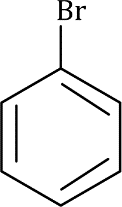
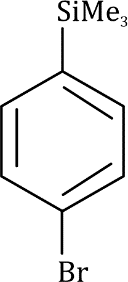
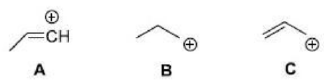
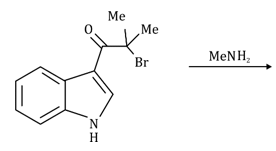
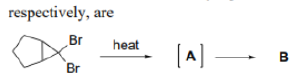
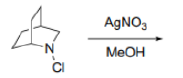
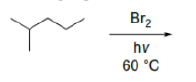
Correct option is A
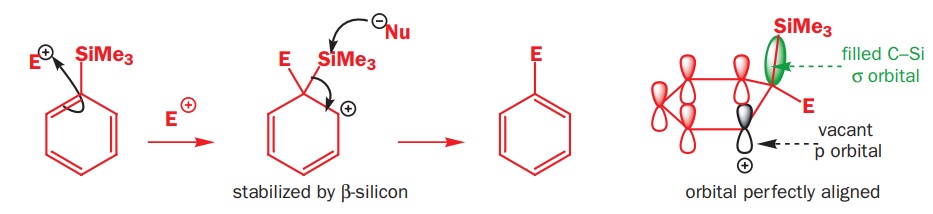
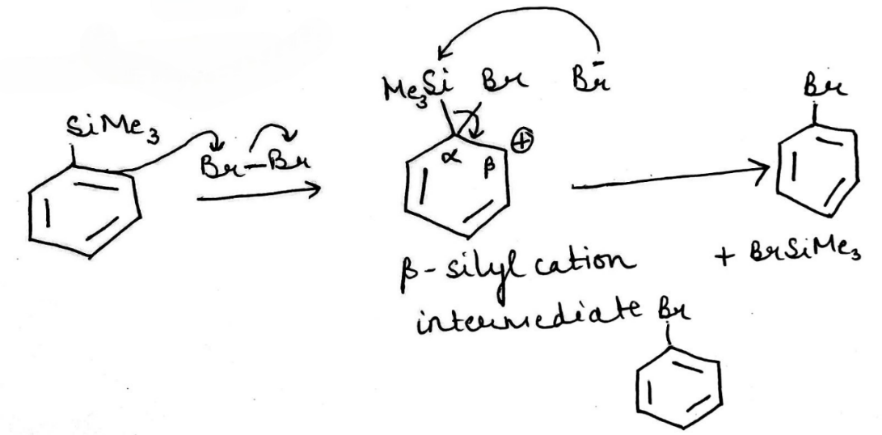
Trimethylsilyl and other silyl groups stabilize a positive charge on a β carbon and are lost very easily. They can be thought of as very reactive protons or ‘super protons’
The silyl group is replaced by the electrophile at the same atom on the ring-this is known as ipso substitution. The electrophile reacts to produce the most stable cation-in this case β to silicon. Cleavage of the weakened C–Si bond by any nucleophile leads directly to the ipso product. The Latin word ipso means ‘the same’—the same site as that occupied by the SiR3 group.