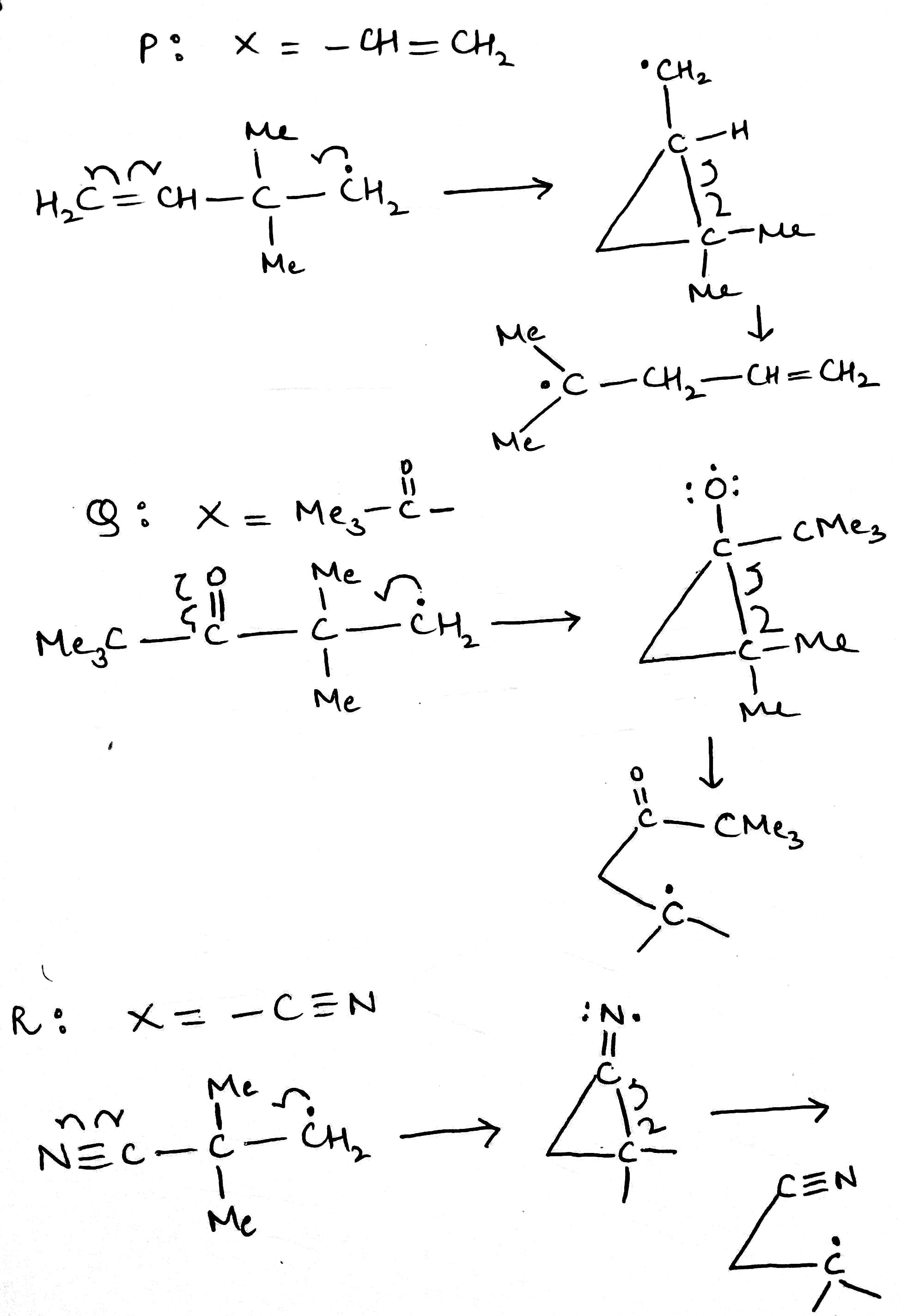
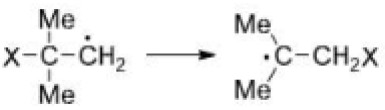
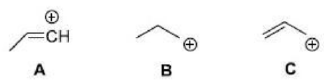
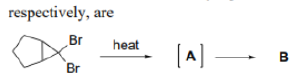
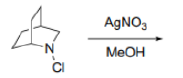
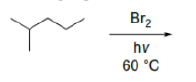
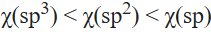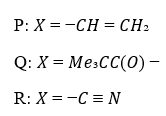Correct option is A
The radical will be more stable on a less electronegative atom. Also, the nitrogen atom is sp hybridized (one lone pair and one sigma bond) while the oxygen atom is sp2 hybridized (two lone pairs and one sigma bond). Since the s character is more on N, the radical will be least stable on it.
The electronegativity of an atom changes depending on the hybridization of the orbital employed in bonding. Electrons in s orbitals are held more tightly than electrons in p orbitals. Hence, a bond to an atom that employs an spx hybrid orbital for bonding will be more heavily polarized to that atom when the hybrid orbital has more s character. That is, when electronegativities are compared for different hybridization schemes of a given element, the order