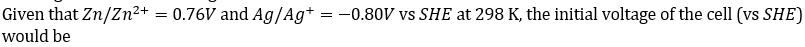Correct option is D
The Nernst equation is a chemical thermodynamical relationship that permits the calculation of the reduction potential of a reaction (half-cell or full cell reaction) from the standard electrode potential, absolute temperature, the number of electrons involved in the redox reaction, and activities (often approximated by concentrations) of the chemical species undergoing reduction and oxidation respectively.

The reaction quotient (Qr), also often called the ion activity product (IAP), is the ratio between the chemical activities (a) of the reduced form (the reductant ,aRed) and the oxidized form (the oxidant ,aOx). The chemical activity of a dissolved species corresponds to its true thermodynamic concentration taking into account the electrical interactions between all ions present in solution at elevated concentrations. For a given dissolved species, its chemical activity (a) is the product of its activity coefficient (γ) by its molar(mol/L solution), or molal (mol/kg water),concentration (C): a = γ C. So, if the concentration (C, also denoted here below with square brackets [ ]) of all the dissolved species of interest are sufficiently low and that their activity coefficients are close to unity, their chemical activities can be approximated by their concentrations as commonly done when simplifying, or idealizing, a reaction for didactic purposes:
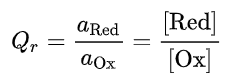
At chemical equilibrium, the ratio Qr of the activity of the reaction product (aRed) by the reagent activity (aOx) is equal to the equilibrium constant K of the half-reaction:
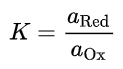

where Qris the reaction quotient and R is the
universal ideal gas constant. The cell potential E associated with the electrochemical reaction is defined as the decrease in Gibbs free energy per coulomb of charge transferred, which leads to the relationship
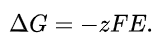
The constant F (the Faraday constant) is a unit conversion factor F=NAq, where NAis the Avogadro constant and q is the fundamental electron charge. This immediately leads to the Nernst equation, which for an electrochemical half-cell is