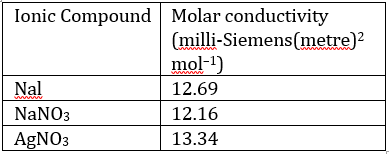
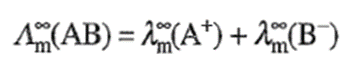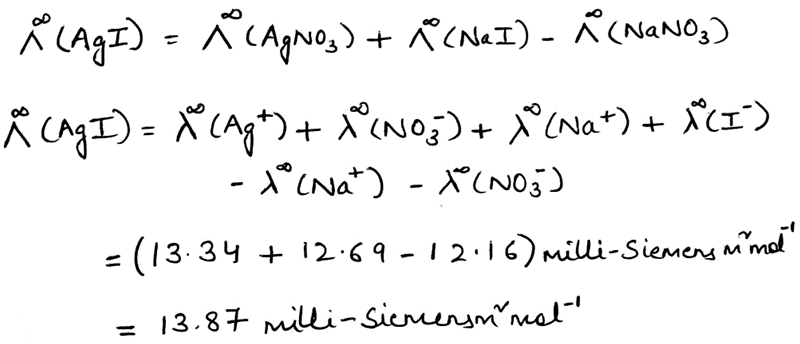Correct option is A
For a strong electrolyte, the value of molar conductivity
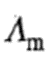
in a very dilute solution, say in 0.001 or 0.000 1 M solution, is very close to the limiting value of the conductivity
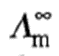
at infinite dilution (or at zero concentration obtained by extrapolation). On the other hand, the corresponding value for a weak electrolyte is very far away from the limiting value at zero concentration.

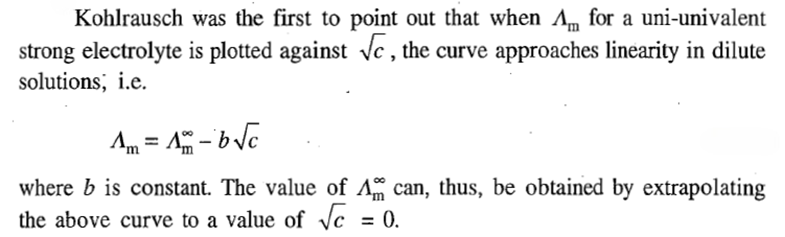
The extrapolation method cannot be employed for a weak electrolyte as
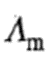
versus (c)1/2 curve does not approach linearity in solutions as dilute as 0.0001M. In fact, the variation of
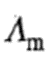
with dilution is very rapid. However, the value of
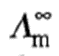
for a weak electrolyte can be determined by the application of Kohlrausch's law of independent migration of ions. This law states that at infinite dilution, where dissociation for all electrolytes is complete (including weak electrolytes since α → 1 as c → 0; Ostwald dilution law) and where all interionic effects disappear (because of larger distance between ions), each ion migrates independently of its co-ion and contributes to the total molar conductivity of an electrolyte a definite share which depends only on its own nature and not at all on the ion with which it is associated. Thus,
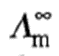
of the electrolyte must be equal to the sum of the molar conductivities of the ions composing it. Thus