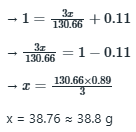Correct option is A
The ionic strength of a solution is a measure of the concentration of ions in that solution. Ionic compounds, when dissolved in water, dissociate into ions. The total electrolyte concentration in solution will affect important properties such as the dissociation constant or the solubility of different salts. One of the main characteristics of a solution with dissolved ions is the ionic strength. Ionic strength can be molar (mol/L solution) or molal (mol/kg solvent) and to avoid confusion the units should be stated explicitly.
The molar ionic strength, I, of a solution is a function of the concentration of all ions present in that solution.
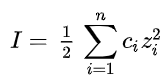
where one half is because we are including both cations and anions, ci is the molar concentration of ion i (M, mol/L), zi is the charge number of that ion, and the sum is taken over all ions in the solution.
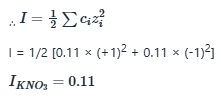
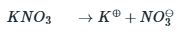
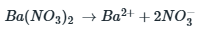
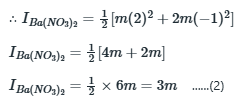

The value of x or the amount of
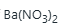
added should raise the ionic strength to 1.