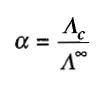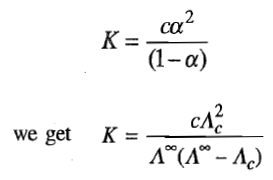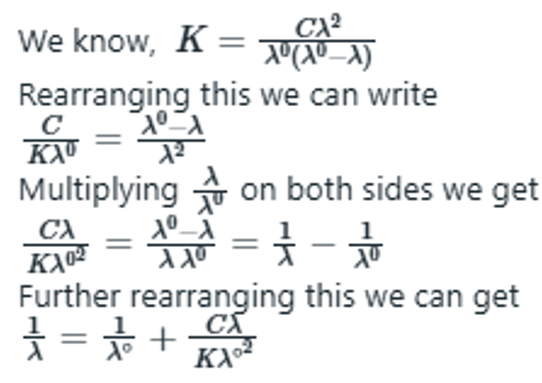Correct option is B
According to the Arrhenius theory of electrolytic dissociation, there exists an equilibrium between the undissociated molecule AB and the ions
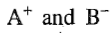
which result from the dissociation of the molecule. Thus,
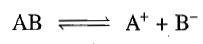
This equilibrium is characterized by the equilibrium constant, defined as
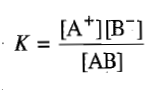

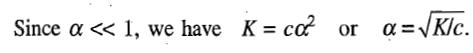

according to which the degree of dissociation increases as c decreases.
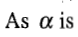
increased, the concentrations of the ions
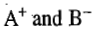
are also increased. Thus, the increase of conductance for a weak electrolyte is primarily due to the increase in the number of ions on dilution.
Expression of degree of dissociation
Arrhenius suggested that the degree of dissociation of an electrolyte can be calculated using the expression