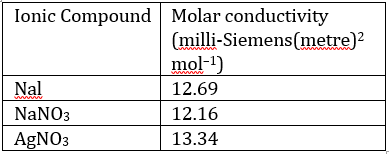
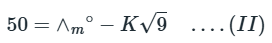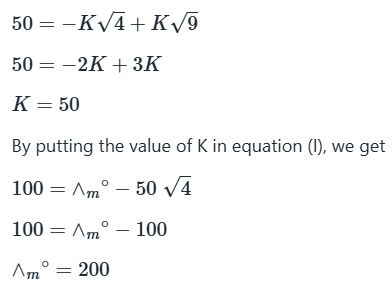Correct option is B
The molar conductivity of an electrolyte solution is defined as its conductivity divided by its molar concentration:
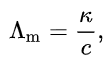
where
κ is the measured conductivity (formerly known as specific conductance),
c is the molar concentration of the electrolyte.
The SI unit of molar conductivity is siemens metres squared per mole (S m2 mol-1).However, values are often quoted in S cm2 mol-1. In these last units, the value of Λm may be understood as the conductance of a volume of solution between parallel plate electrodes one centimeter apart and of sufficient area so that the solution contains exactly one mole of electrolyte.
There are two types of electrolytes: strong and weak. Strong electrolytes usually undergo complete ionization, and therefore they have higher conductivity than weak electrolytes, which undergo only partial ionization. For strong electrolytes, such as salts, strong acids and strong bases, the molar conductivity depends only weakly on concentration. On dilution there is a regular increase in the molar conductivity of strong electrolyte, due to the decrease in solute–solute interaction. Based on experimental data Friedrich Kohlrausch proposed the non-linear law for strong electrolytes:
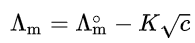
where
Λ∘m is the molar conductivity at infinite dilution (or limiting molar conductivity), which can be determined by extrapolation of Λm as a function of √c,
K is the Kohlrausch coefficient, which depends mainly on the stoichiometry of the specific salt in solution