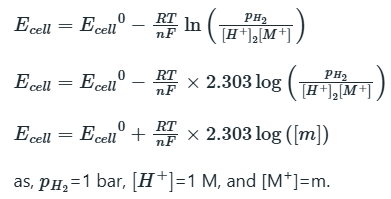Correct option is A
The Nernst equation is a chemical thermodynamical relationship that permits the calculation of the reduction potential of a reaction (half-cell or full cell reaction) from the standard electrode potential, absolute temperature, the number of electrons involved in the redox reaction, and activities (often approximated by concentrations) of the chemical species undergoing reduction and oxidation respectively.
The standard half cell potential of the reaction
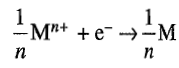
is a measure of reduction tendency of
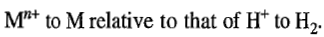
The standard potential of the latter reaction
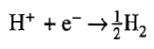
is taken to be zero (reference level).