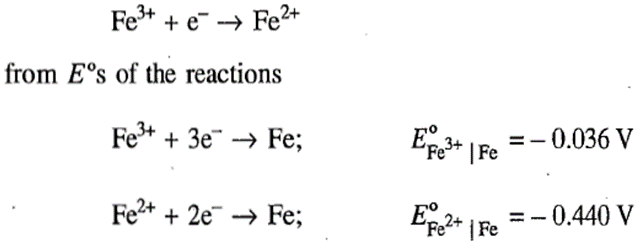Correct option is D
A given reaction may be obtained from other reactions by carrying out the simple mathematical manipulations such as addition, subtraction, multiplication by a number and so on. Since G° is a state function, the ΔG° of the given reaction may be obtained by carrying out the corresponding manipulations on ΔG°s values of the reactions. This procedure, however, cannot be adopted while calculating E° of a half-cell reaction from the values of other half-cell reactions. Nevertheless, we can determine the potential of a half-cell reaction from the potentials of other half-cell reactions via the associated ΔG° values. First of all, we write

for each and every half-cell reaction and then carry out the appropriate manipulations of the reactions along with the corresponding ΔG°s values to give the given half-cell reaction and its ΔG° value. Knowing the number n of electrons involved in the given reaction and the obtained value of ΔG°, the value E° may be determined. To illustrate the procedure, we compute E° for the reaction