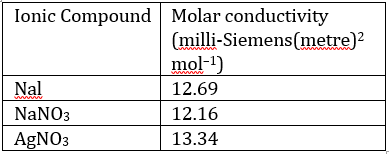
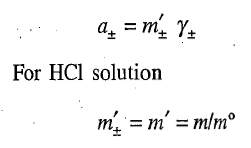Correct option is D
The standard half-cell potentials are employed for calculating accurate values of various quantities such as equilibrium constant of a weak acid and ionic product of water. It is, therefore, essential to know their accurate values. For this purpose, the method of extrapolation is employed. To illustrate the method, we consider the following cell.
Pt∣H2(1bar)∣HCl(a)∣AgCl(s)∣AgThe cell reaction isAgCl+21H2→Ag+Cl−+H+
and the cell potential is given by
E=Eo−FRTln((fH2fo)1/2(aH+)(aCl−))
Assuming ideal behaviour for the gas, we get
E=Eo−FRTln((aH+)(aCl−))
Since individual ionic activities cannot be determined, we express
a±2=(aH+)(aCl−)
Now the mean activity may be expressed in terms of mean molality m± of the ions and the mean activity coefficient γ± by the relation
where m is the molality of the solution