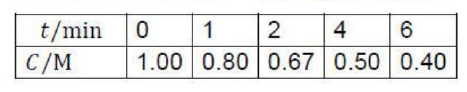
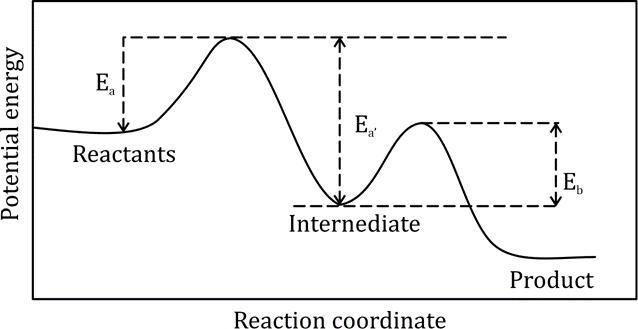
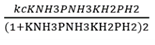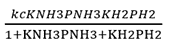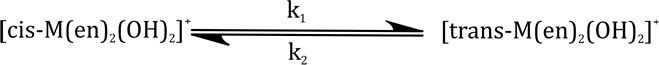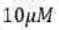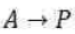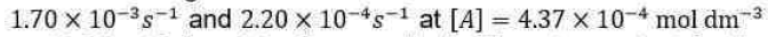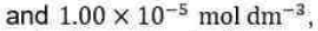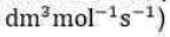Correct option is D
Langmuir proposed the theory of adsorption of a gas on the surface of a solid. He considered the surface of the solid to be made up of elementary sites each of which could adsorb one gas molecule. It is assumed that all adsorption sites are equivalent and the ability of the gas molecule to get bound to any one site is independent of whether or not the neighboring sites are occupied. It is further assumed that a dynamic equilibrium exists between the adsorbed molecules and the free molecules. If A is the gas molecule and M is the surface site, then

where ka and kd are the rate constants for adsorption and desorption, respectively. The rate of adsorption is proportional to the pressure of A, viz., PA and the number of vacant sites on the surface, viz., N(1-ϴ) where N is the total number of sites and ϴ is the fraction of surface sites occupied by the gas molecules, i.e.