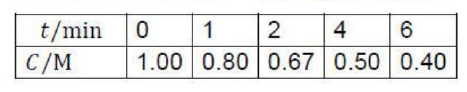
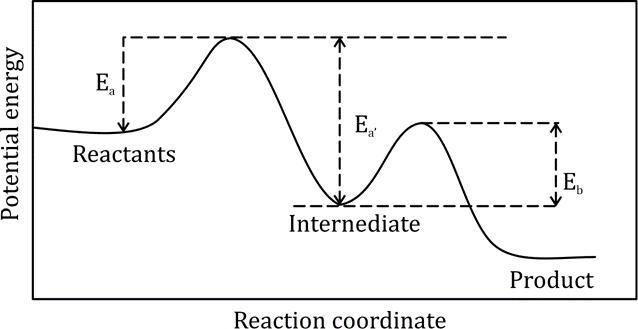
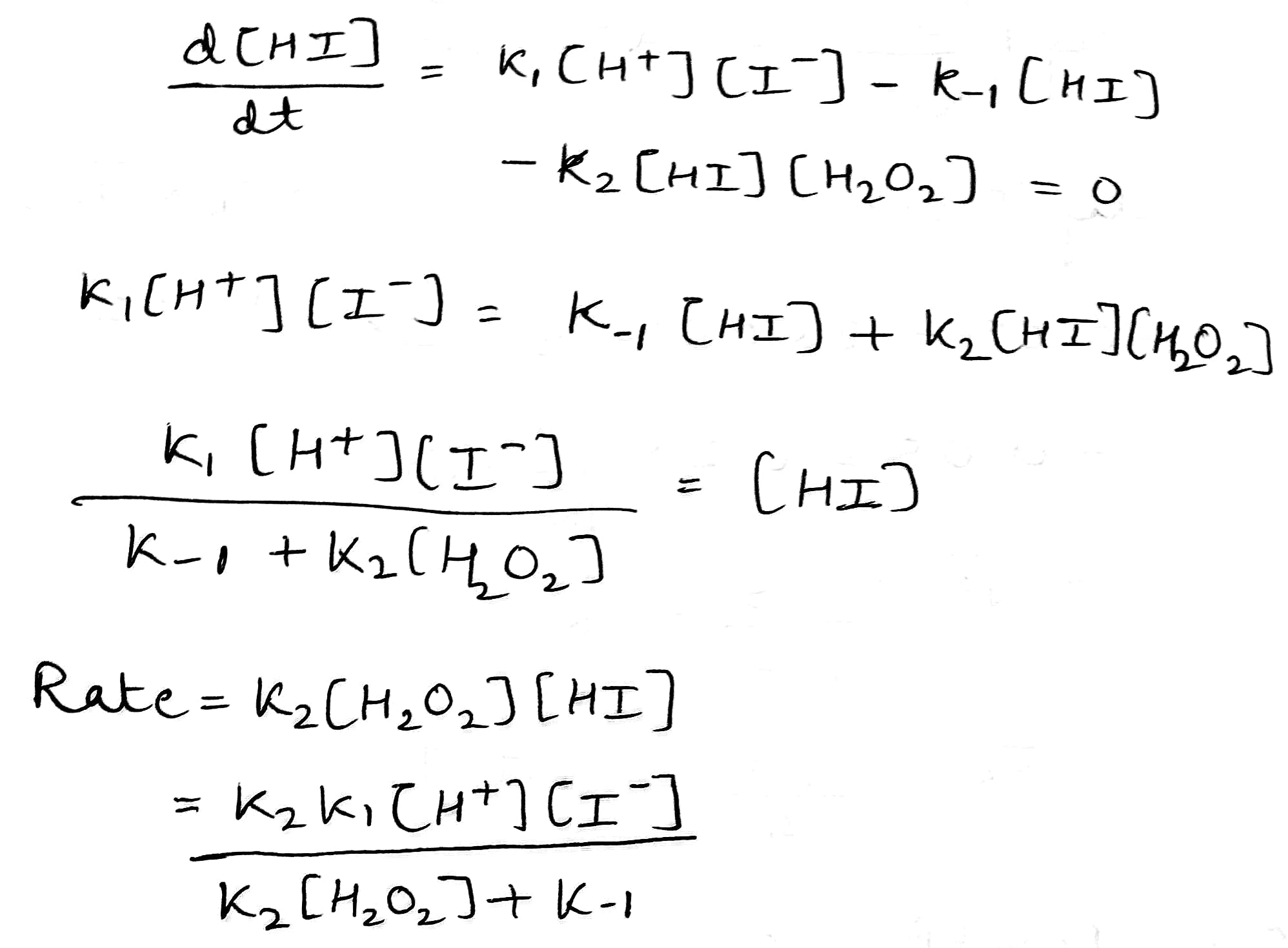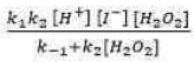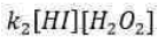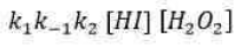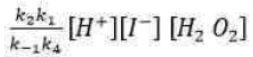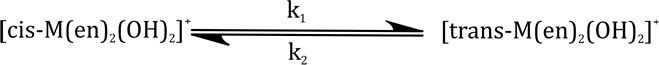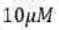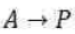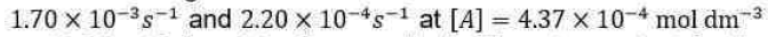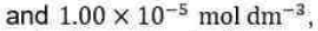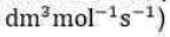Correct option is A
Consecutive or Sequential reactions
In many cases the product formed in one of the elementary reactions acts as the reactant for some other elementary reaction.

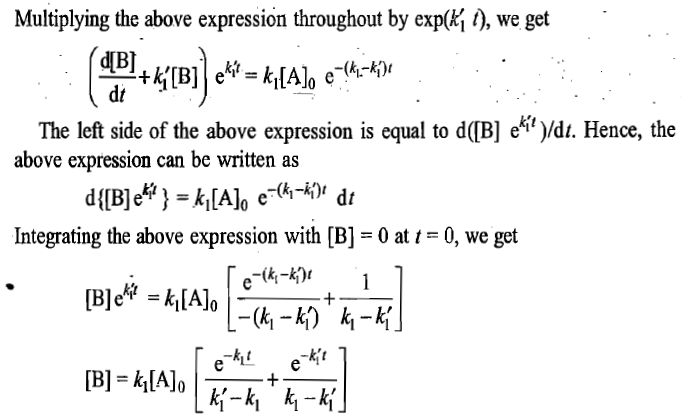
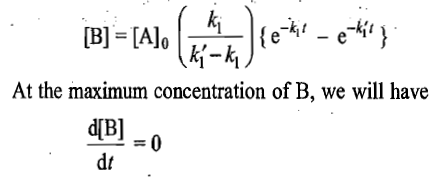
Steady-State Approximation
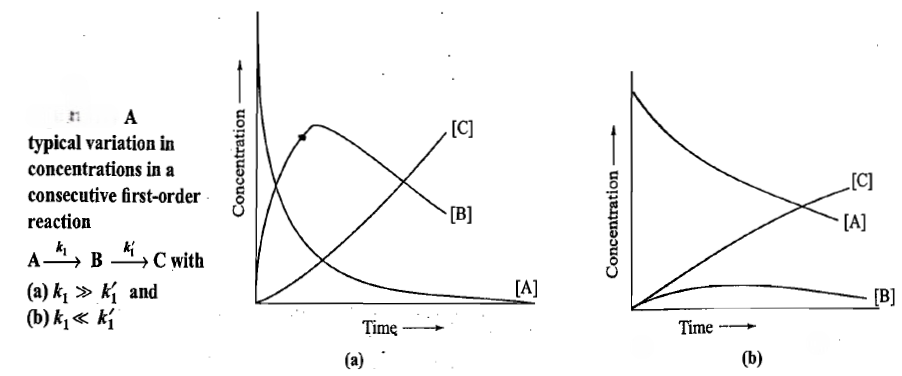
Figures a and b illustrate the typical variations in the concentrations of A,B and C for the two consecutive first-order reactions with
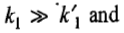
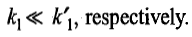
In figure a, the concentration of A disappears very rapidly as
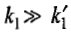
and since the rate at which B disappears is relatively slow, the concentration of B increases rapidly and after attaining the maximum in a small time interval, starts decreasing.
In figure b, the concentration of A decreases slowly as the reaction rate constant k1 has a small value. The concentration of B practically remains constant for a reasonable length of time during the reaction, except at the very beginning and at the end. This constancy results from the fact that the intermediate B is very reactive as its transformation to C occurs at a very fast rate. In fact, the concentration of B at any instant will be much smaller than that of A.