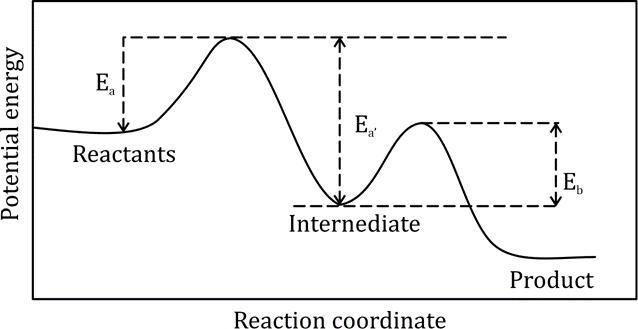
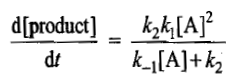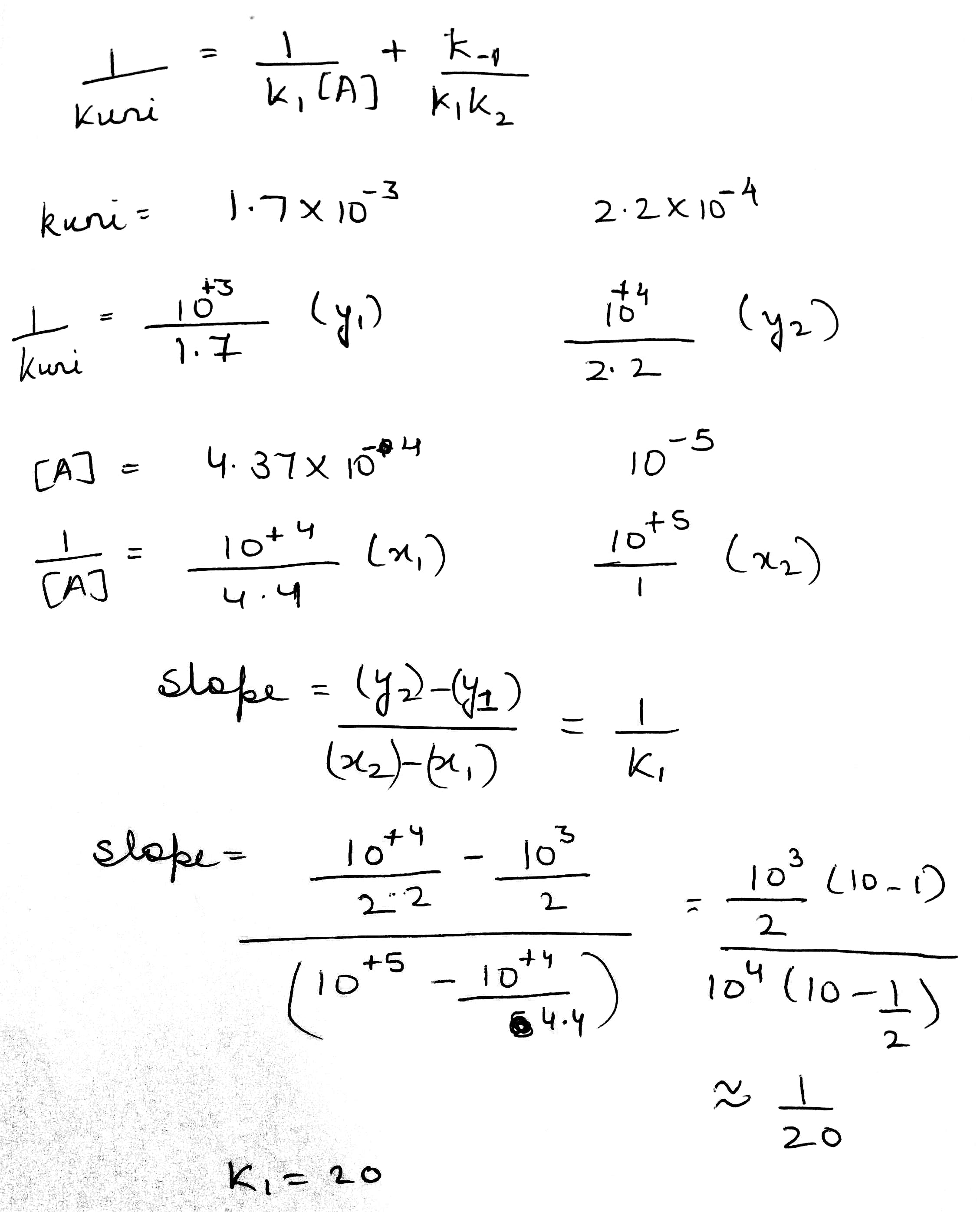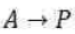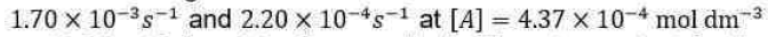Correct option is D
Many thermal decomposition and isomerization reactions follow the mechanism given below:
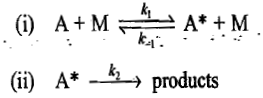
where A represents the molecule undergoing the thermal decomposition or isomerization reaction, and M represents any other molecule. Step (i) represents an equilibrium reaction. The forward reaction represents a collision of molecule A with a molecule of M and thereby the molecule A is activated. The backward reaction represents the deactivation of A molecule. Step (ii) represents the decomposition of the activated A molecule. Two simple cases may be distinguished.


Lindemann mechanism for the first order reactions
The products are formed only when reactant molecules come close and collide together at one and the same time. During the collision, molecular rearrangement takes place which leads to the formation of products. For example, a second order reaction of the types
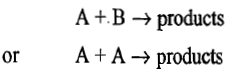
may be traced to a bimolecular collision process involving A and B, and A and A, respectively. However, the first order reactions in the gas phase cannot be accounted for by the above collision theory. The first successful explanation of first order reactions was provided by Lindemann. The mechanism is
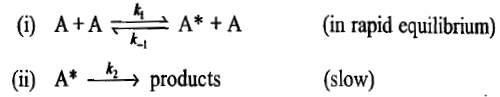
Step (i) involves a rapid equilibrium reaction where in the forward reaction, a molecule A is activated by colliding with another A molecule and in the backward reaction, the excited molecule A is deactivated by colliding with another A molecule. Step (ii) represents another possibility of deactivation where the excited molecule is decomposed to give the products. Since the step (ii) is slow and rate determining, the rate at which products are formed will be determined by the unimolecular decay and hence first-order kinetics will be observed.
The general rate law in the present case becomes