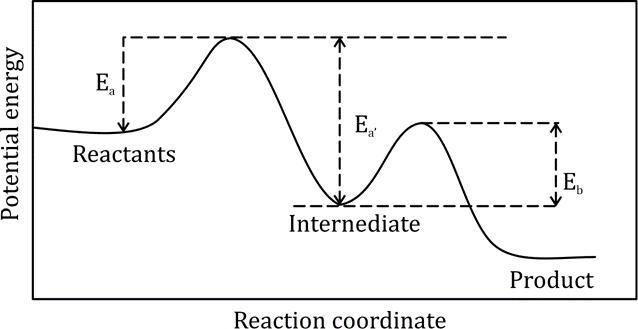
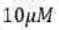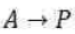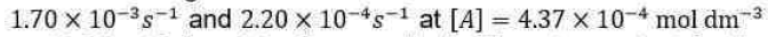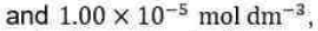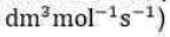Correct option is B
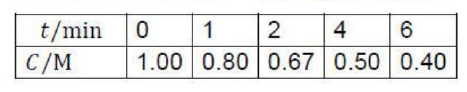
The reaction rate or rate of reaction is the speed at which a chemical reaction takes place, defined as proportional to the increase in the concentration of a product per unit time and to the decrease in the concentration of a reactant per unit time. Reaction rates can vary dramatically. For most reactions, the rate decreases as the reaction proceeds. A reaction's rate can be determined by measuring the changes in concentration over time.
For a chemical reaction aA + bB → pP + qQ, the rate equation or rate law is a mathematical expression used in chemical kinetics to link the rate of a reaction to the concentration of each reactant. For a closed system at constant volume, this is often of the form
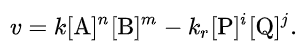
For reactions that go to completion (which implies very small kr), or if only the initial rate is analyzed (with initial vanishing product concentrations), this simplifies to the commonly quoted form
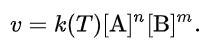
In these equations k(T) is the reaction rate coefficient or rate constant, although it is not really a constant, because it includes all the parameters that affect reaction rate, except for time and concentration. The exponents n and m are called reaction orders and depend on the reaction mechanism. For an elementary (single-step) reaction, the order with respect to each reactant is equal to its stoichiometric coefficient. For complex (multistep) reactions, however, this is often not true and the rate equation is determined by the detailed mechanism.
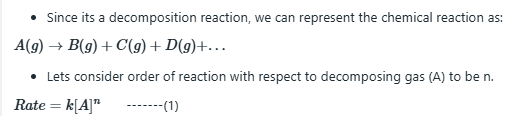
Case 1: 10% of gas reacted
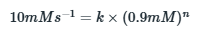
Case 2: 40% of gas reacted