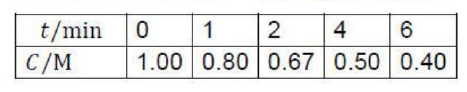
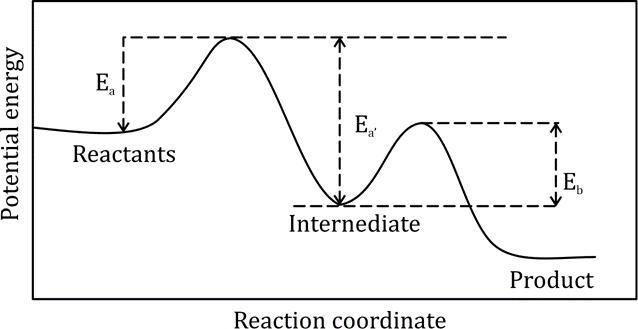
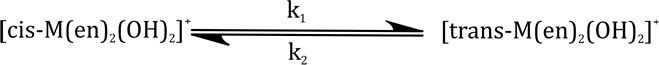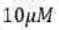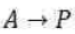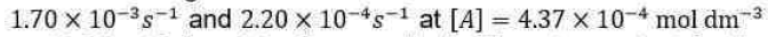Correct option is B
The dependence of rate constant on temperature may be derived from the Van't Hoff equation as applicable to a reaction at equilibrium. Consider a general equilibrium reaction

where ΔE is the energy change of the reaction
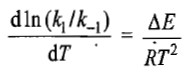
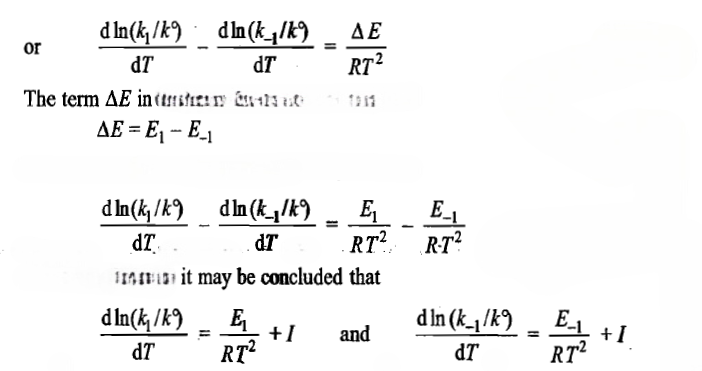
where I represents a constant which may have any value including a value of zero. If we take it to be zero, then the above two relations may be written as

The constant A is frequently referred to as the pre-exponential factor.
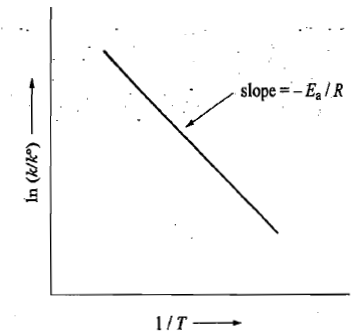
The constant Ea is known as the energy of activation.