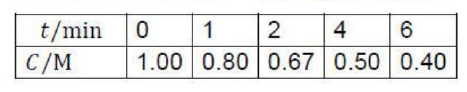
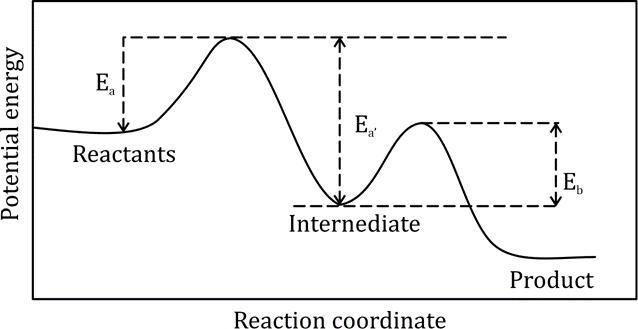
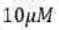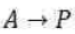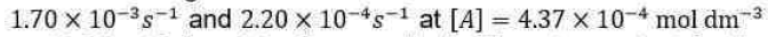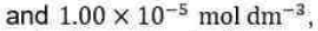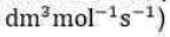Correct option is A
It is found experimentally for many reactions that a plot of ln k against 1/T gives a straight line. This behaviour is normally expressed mathematically by introducing two parameters, one representing the intercept and the other the slope of the straight line, and writing the Arrhenius equation
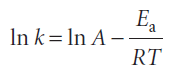
The parameter A, which corresponds to the intercept of the line at 1/T = 0, is called the pre-exponential factor or the ‘frequency factor’. The parameter Ea, which is obtained from the slope of the line
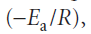
is called the activation energy. Collectively the two quantities are called the Arrhenius parameters
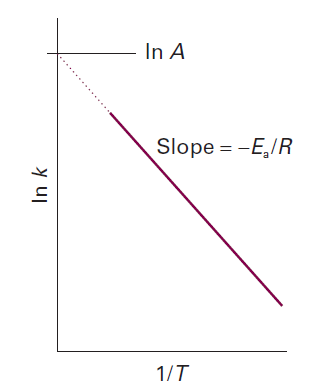
The fact that Ea is given by the slope of the plot of ln k against 1/T means that, the higher the activation energy, the stronger the temperature dependence of the rate constant (that is, the steeper the slope). A high activation energy signifies that the rate constant depends strongly on temperature. If a reaction has zero activation energy, its rate is independent of temperature. In some cases the activation energy is negative, which indicates that the rate decreases as the temperature is raised.