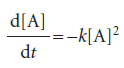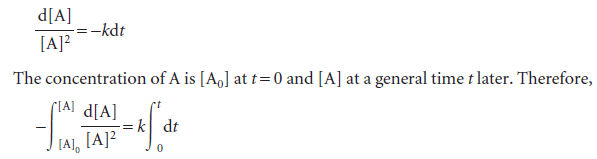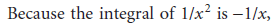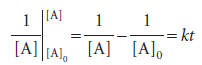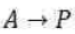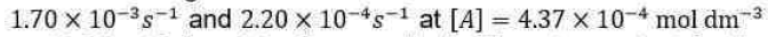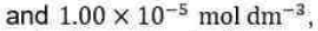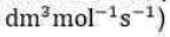Correct option is C
The definition of rate
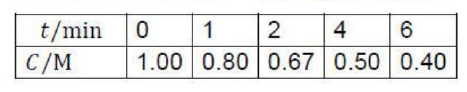
Consider a reaction of the form A + 2 B →3 C + D, in which at some instant the molar concentration of a participant J is [J] and the volume of the system is constant. The instantaneous rate of consumption of one of the reactants at a given time is −d[R]/dt, where R is A or B. This rate is a positive quantity. The rate of formation of one of the products (C or D, which we denote P) is d[P]/dt (note the difference in sign). This rate is also positive. It follows from the stoichiometry for the reaction A + 2 B →3 C + D that
so there are several rates connected with the reaction.
Rate laws and rate constant
The rate of reaction is often found to be proportional to the concentrations of the reactants raised to a power. For example, the rate of a reaction may be proportional to the molar concentrations of two reactants A and B, so we write
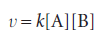
with each concentration raised to the first power. The coefficient k is called the rate constant for the reaction. The rate constant is independent of the concentrations but depends on the temperature. An experimentally determined equation of this kind is called the rate law of the reaction. More formally, a rate law is an equation that expresses the rate of reaction as a function of the concentrations of all the species present in the overall chemical equation for the reaction at some time:
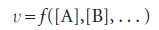
Reaction order
Many reactions are found to have rate laws of the form
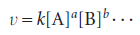
The power to which the concentration of a species (a product or a reactant) is raised in a rate law of this kind is the order of the reaction with respect to that species.
Second-order reactions