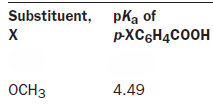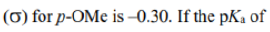Correct option is D
What we would ideally like to do is find a way to quantify the effects that electron-donating or -withdrawing groups have on the transition state or intermediate during the course of a reaction. This will then give us an idea of what the transition state is really like. The first question is: can we define exactly how efficient a given group is at donating or withdrawing electrons? Hammett took the arbitrary decision to use the pKa of an acid as a guide.
Hammett decided not to use the pKas themselves for his correlation but defined a new parameter, which he called σ. This σ shows how electron-donating or -withdrawing a group is relative to H as a ratio of the logKas or the difference of the pKas between the substituent and benzoic acid itself. If σ = 0 the substituent has no effect: it is electronically the same as H. If σ is positive, the substituent is electron withdrawing. This is unfortunate perhaps, but just remember that the comparison is with acid strength. Positive σ means a stronger acid so the substituent is electron withdrawing. The more positive the charge induced on the ring by a substituent, the larger its σ value. Negative σ means weaker acid and electron donation.
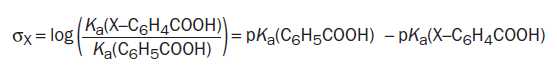
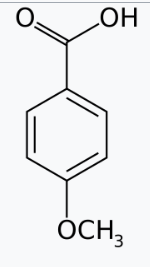
In case of p-anisic acid, the substituent (-OCH3) is electron-donating and the anions of the benzoic acids are correspondingly less stable so these are the weakest acids.