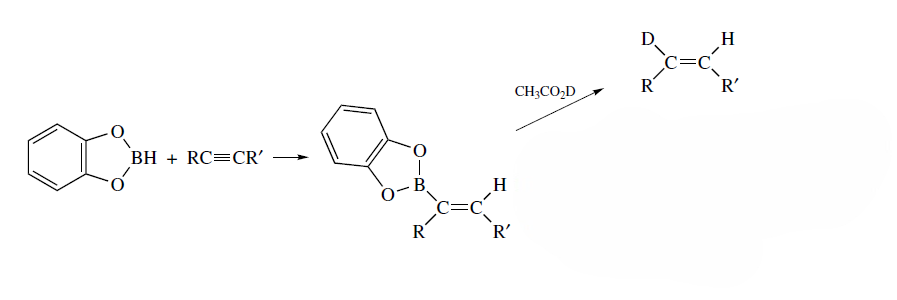
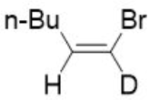
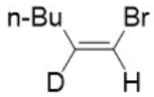
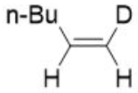
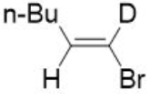
Correct option is A
Hydroboration
Borane, BH3, is an avid electron-pair acceptor, having only six valence electrons on boron. Pure borane exists as a dimer in which two hydrogens bridge the borons. In aprotic solvents that can act as electron donors such as ethers, tertiary amines, and sulfides, borane forms Lewis acid-base adducts. Borane dissolved in THF or dimethyl sulfide undergoes addition reactions rapidly with most alkenes. This reaction, which is known as hydroboration, has been extensively studied. Hydroboration is highly regioselective and is stereospecific. The boron becomes bonded primarily to the less substituted carbon atom of the alkene. A combination of steric and electronic effects work together to favor this orientation. Borane is an electrophilic reagent.
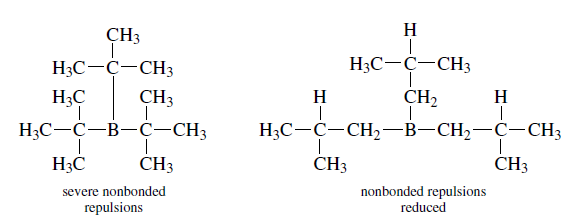
The structure of dicyclohexyl borane (Cy2BH) is shown below
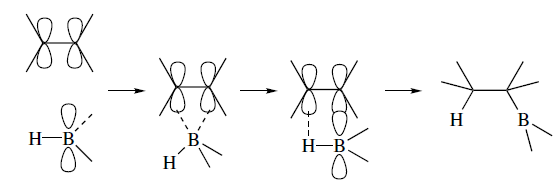
Hydroboration is a stereospecific syn addition. The addition occurs through a four-centre transition state with essentially simultaneous bonding to boron and hydrogen. Both the new C-B and C-H bonds are, therefore formed from the same side of the double bond. In molecular orbital terms, the addition is viewed as taking place by interaction of the filled alkene pi orbital with the empty p orbital on boron, accompanied by concerted C-H bond formation.
There is a preference for approach of the borane from the less hindered side of the molecule.
Hydroboration of alkynes: Alkynes are reactive toward hydroboration reagents. Catecholborane is a particularly useful reagent for hydroboration of alkynes.