Correct option is C
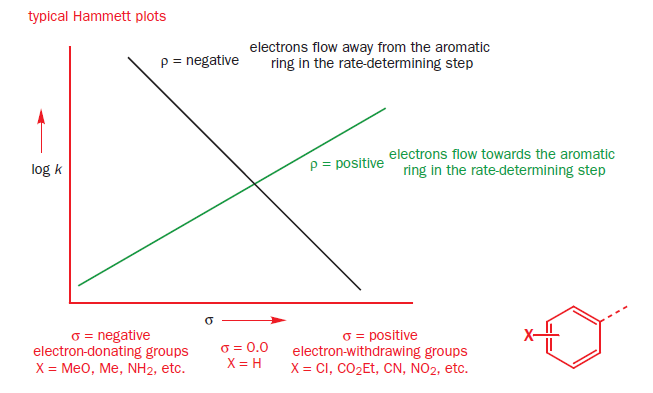
In organic chemistry, the Hammett equation describes a linear free-energy relationship relating reaction rates and equilibrium constants for many reactions involving benzoic acid derivatives with meta- and para-substituents to each other with just two parameters: a substituent constant and a reaction constant.
= Reference constant
= Substituent constant
= Reaction rate constant
relating the equilibrium constant, K, for a given equilibrium reaction with substituent R and the reference constant K0 when R is a hydrogen atom to the substituent constant which depends only on the specific substituent R and the reaction rate constant which depends only on the type of reaction but not on the substituent used.
The Hammett reaction constant ρ measures the sensitivity of the reaction to electronic effects. A positive ρ value means more electrons in the transition state than in the starting material. A negative ρ value means fewer electrons in the transition state than in the starting material.
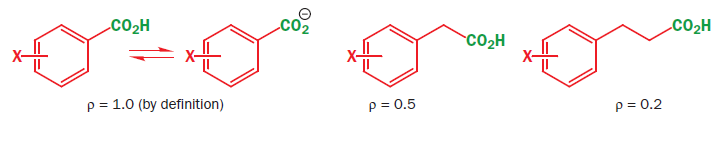
If we simply move the carboxylic acid away from the ring, the ρ value for ionization gets less. This is just the effect of a more distant substituent. When there are two saturated carbons between the benzene ring and the carboxylic acid, there is almost no effect. When we are using the aromatic ring as a probe for a reaction mechanism, it must be placed not too far away from the reaction centre. However, if we restore electronic communications with a double bond, ρ goes back up again to a useful value.