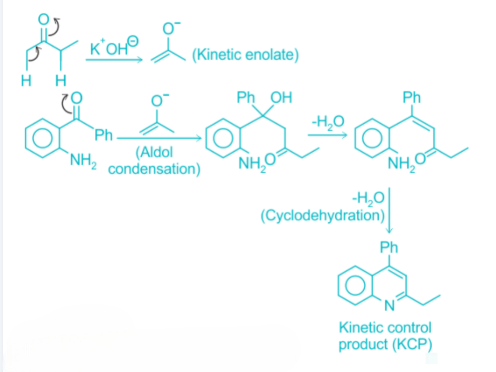
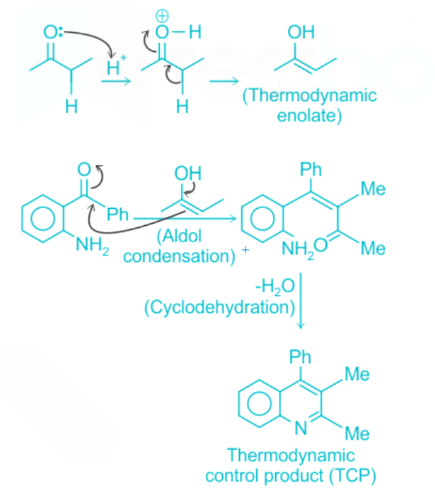
Correct option is B
Consider the addition of HCN to butenone. Two products can be formed.
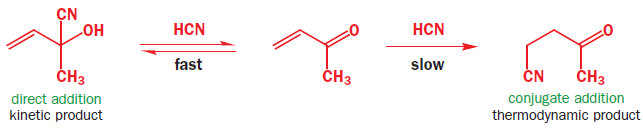
The ‘direct’ addition to the left means that cyanide ion must attack the carbonyl group directly while the ‘conjugate’ addition to the right means that it must attack the less electrophilic alkene. The second is a slower reaction but gives the more stable product. Both reactions have an alkoxide anion as an intermediate.
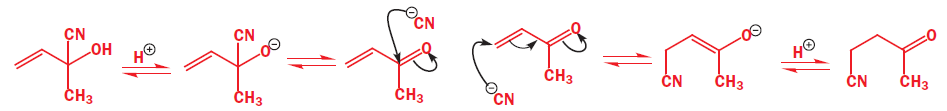
The energy profile diagram for these two reactions is quite complicated. It has the starting material in the middle, as in the mechanism above, and so extent of reaction increases both to the right for thermodynamic control and to the left for kinetic control.
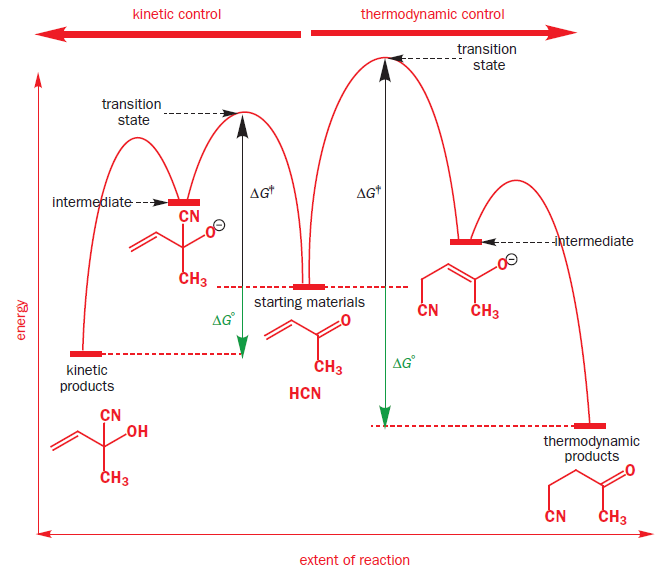
At low temperature, the reaction is under kinetic control (rate, irreversible conditions) and the major product is that from the fastest reaction.
At high temperature, the reaction is under thermodynamic control (equilibrium, reversible conditions) and the major product is the more stable system.