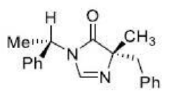
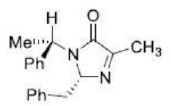
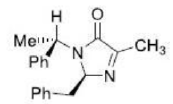
Correct option is A
A nucleophilic substitution reaction is a class of chemical reactions in which an electron-rich chemical species or nucleophile reacts with another electron-deficient molecule and replaces a functional group within. The molecule that contains the electrophile and the leaving group is known as substrate. Lithium disopropylamide or LDA is a chemical compound with the molecular formula LiN(CH(CHs)2)2. It is a strong base and has good solubility in non-polar organic solvents and LDA is non-nucleophilic.
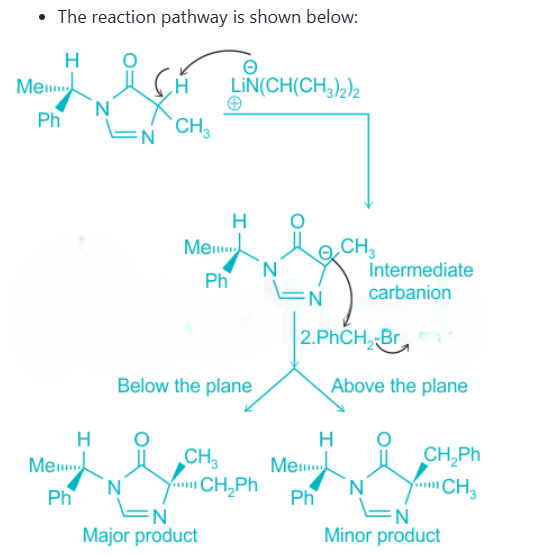
From the above reaction, we can see that LDA act as a strong base and abstract a proton from the substrate in the first step of the reaction. The intermediate carbanion will act as a nucleophile and react with PhCh2Br (Electrophile) via a nucleophilic substitution reaction. The reaction can lead to two products depending upon the streic effect experienced by the incoming group with the substituent presents in intermediate carbanion. The phenyl group (-Ph) being a bulky substituent will cause more steric hindrance than -Me group. Also, the incoming electrophile is a much bulkier substituent than the methyl group. The bulky phenyl group (-Ph) is present above the plane in the intermediate carbanion. This indicates that the incoming group (-CHzPh) will face more steric effect when it will be placed in above the plane. Thus, in the major product incoming electrophile will be placed below the plane.