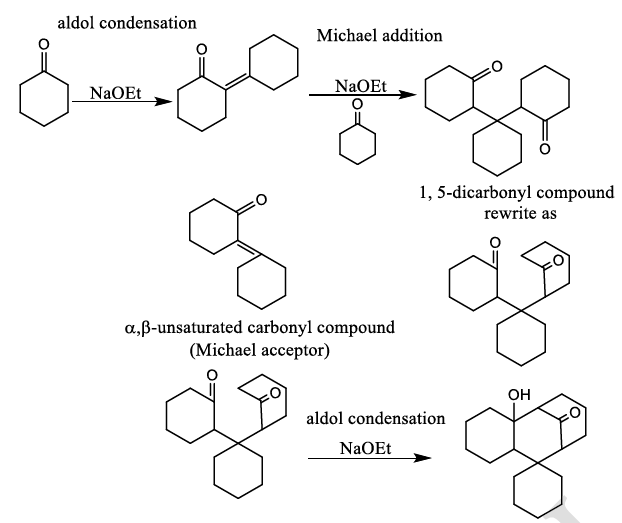
Correct option is A
An aldol condensation is a condensation reaction in organic chemistry in which two carbonyl moieties (of aldehydes or ketones) react to form a β-hydroxyaldehyde or β-hydroxyketone (an aldol reaction), and this is then followed by dehydration to give a conjugated enone. The overall reaction equation is as follows (where the Rs can be H)
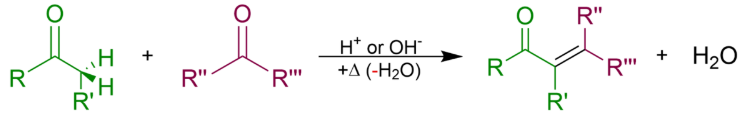
The mechanism for base-catalyzed aldol condensation can be seen in the image below.
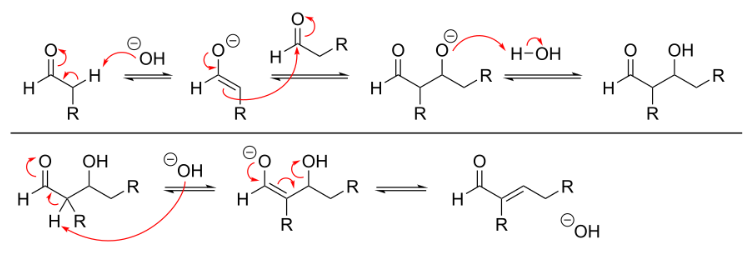
The process begins when a free hydroxide (strong base) strips the highly acidic proton at the alpha carbon of the aldehyde. This deprotonation causes the electrons from the C–H bond to shift and create a new C–C pi bond. The new pi bond then acts as a nucleophile and attacks the remaining aldehyde in the solution, resulting in the formation of a new C–C bond and regeneration of the base catalyst. In the second part of the reaction, the presence of base leads to elimination of water and formation of a new C–C pi bond. The product is referred to as the aldol condensation product.
In organic chemistry, the Michael reaction or Michael 1,4 addition is a reaction between a Michael donor (an enolate or other nucleophile) and a Michael acceptor (usually an α,β-unsaturated carbonyl) to produce a Michael adduct by creating a carbon-carbon bond at the acceptor's β-carbon. It belongs to the larger class of conjugate additions and is widely used for the mild formation of carbon–carbon bonds.
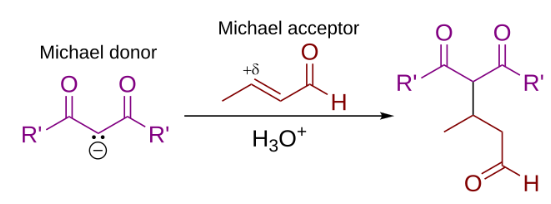
In this general Michael addition scheme, either or both of R and R' on the nucleophile (the Michael donor) represent electron-withdrawing substituents such as acyl, cyano, nitro, or sulfone groups, which make the adjacent methylene hydrogen acidic enough to form a carbanion when reacted with the base, B:. For the alkene (the Michael acceptor), the R" substituent is usually a carbonyl, which makes the compound an α,β-unsaturated carbonyl compound (either an enone or an enal), or R" may be any electron withdrawing group.
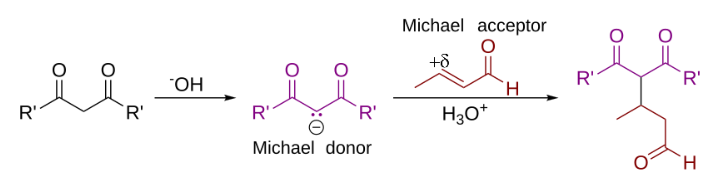
In the reaction mechanism, there is 1 as the nucleophile:
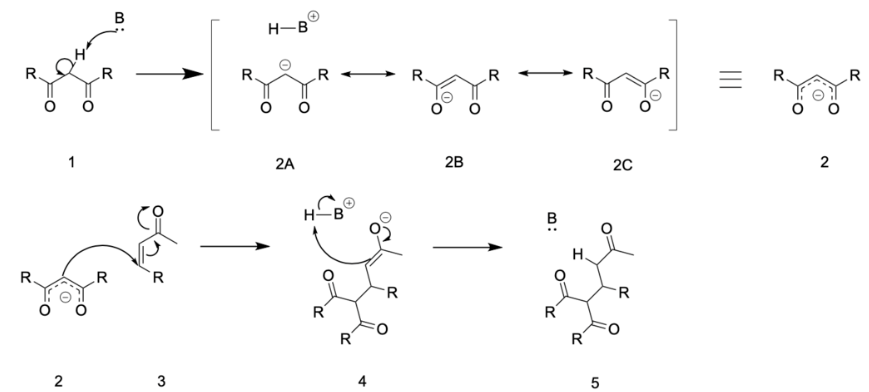
Deprotonation of 1 by a base leads to carbanion 2, stabilized by its electron-withdrawing groups. Structures 2a to 2c are three resonance structures that can be drawn for this species, two of which have enolate ions. This nucleophile reacts with the electrophilic alkene 3 to form 4 in a conjugate addition reaction. Finally, enolate 4 abstracts a proton from protonated base (or solvent) to produce 5.