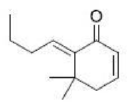
Correct option is B
Wittig reaction
The Wittig reaction or Wittig olefination is a chemical reaction of an aldehyde or ketone with a triphenyl phosphonium ylide called a Wittig reagent. Wittig reactions are most commonly used to convert aldehydes and ketones to alkenes. Most often, the Wittig reaction is used to introduce a methylene group using methylenetriphenylphosphorane (Ph3P=CH2).
Phosphorus atoms, especially those that are positively charged or that carry electronegative substituents, can increase the acidity of protons adjacent to them on the carbon skeleton. Phosphonium salts (made in a manner analogous to the formation of ammonium salts from amines, in other words, by reaction of an alkyl halide with a phosphine) can therefore be deprotonated by a moderately strong base to give a species known as an ylid, carrying (formally) a positive and a negative charge on adjacent atoms. Ylids can alternatively be represented as doubly bonded species, called phosphoranes.
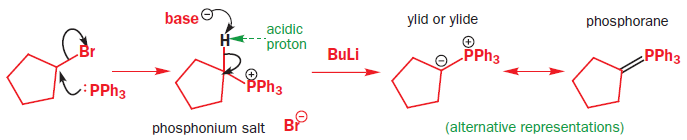
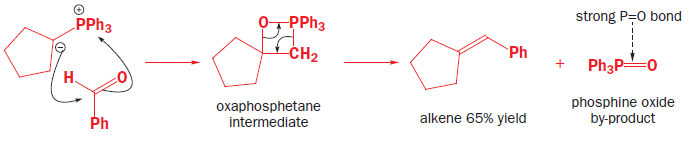
Ylids can be isolated, but are usually used in reactions immediately they are formed. They are nucleophilic species that will attack the carbonyl groups of aldehydes or ketones, generating the four-membered ring oxaphosphetane intermediates. Oxaphosphetanes are unstable: they undergo elimination to give an alkene (65% yield for this particular example) with a phosphine oxide as a byproduct. The phosphorus–oxygen double bond is extremely strong and it is this that drives the whole reaction forward.
With α,β-unsaturated carbonyl compounds such as cyclohexenone it can be deduced from resonance structures that the β position is an electrophilic site which can react with a nucleophile. The negative charge in these structures is stored as an alkoxide anion. Such a nucleophilic addition is called a nucleophilic conjugate addition or 1,4-nucleophilic addition.
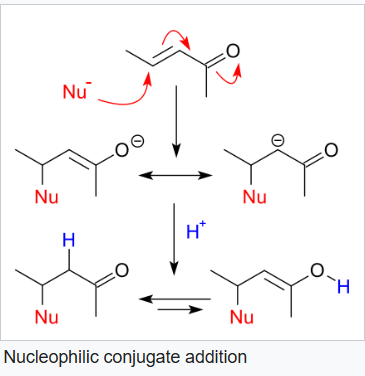
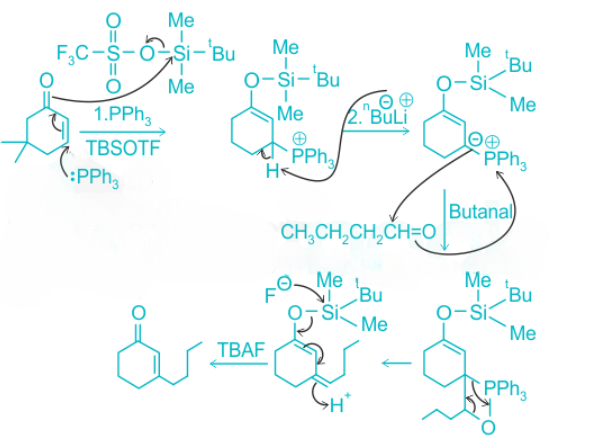
In the above reaction, triphenylphosphine undergoes conjugate addition reaction with the α,β-unsaturated carbonyl compound.
nBuLi (alkyl lithium reagent) abstracts a H+.
The ylide reacts with butanal (Wittig reaction) to give the alkene along with triphenylphosphine oxide. In the last step, TBAF reacts with the tert-butyldimethylsilyl group and deprotects the alcohol group.