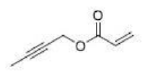
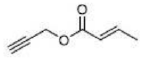
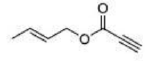
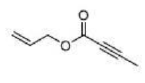
Correct option is A
A cycloaddition is a reaction in which two unsaturated molecules undergo an addition reaction to yield a cyclic product. Formation of cyclic product takes place at the expense of one π (pi) bond in each of the reacting partner and gain of two σ (sigma) bonds at the end of the both components having π ( pi ) bonds. Thus, in this reaction there is loss of two π (pi) bonds of the reactants and gain of two σ (sigma) bonds in the product.
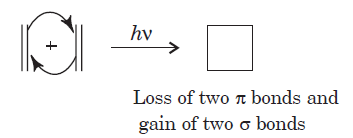
The reaction in the above equation is a [2 + 2] cycloaddition reaction because the reaction involves two electrons from one reacting component and also two electrons from the other.
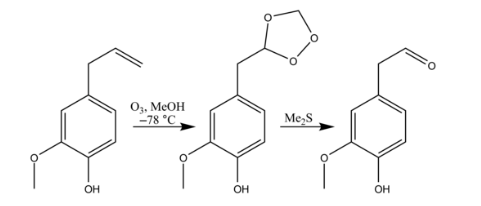
Ozonolysis is an organic reaction where the unsaturated bonds are cleaved with ozone (O3). Multiple carbon–carbon bond are replaced by carbonyl (C=O) groups, such as aldehydes, ketones, and carboxylic acids. The reaction is predominantly applied to alkenes, but alkynes and azo compounds are also susceptible to cleavage. The outcome of the reaction depends on the type of multiple bond being oxidized and the work-up conditions. The use of triphenylphosphine, thiourea, zinc dust, or dimethyl sulfide produces aldehydes or ketones.
An aldol condensation is a condensation reaction in organic chemistry in which two carbonyl moieties (of aldehydes or ketones) react to form a β-hydroxyaldehyde or β-hydroxyketone (an aldol reaction), and this is then followed by dehydration to give a conjugated enone. The overall reaction equation is as follows (where the Rs can be H)
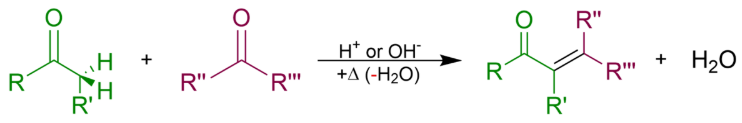
The mechanism for base-catalyzed aldol condensation can be seen in the image below.
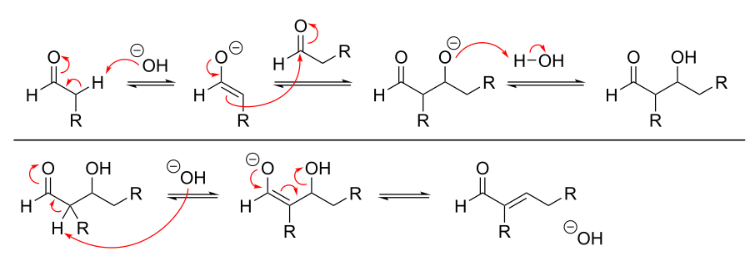
The process begins when a free hydroxide (strong base) strips the highly acidic proton at the alpha carbon of the aldehyde. This deprotonation causes the electrons from the C–H bond to shift and create a new C–C pi bond. The new pi bond then acts as a nucleophile and attacks the remaining aldehyde in the solution, resulting in the formation of a new C–C bond and regeneration of the base catalyst. In the second part of the reaction, the presence of base leads to elimination of water and formation of a new C–C pi bond. The product is referred to as the aldol condensation product.