Correct option is B



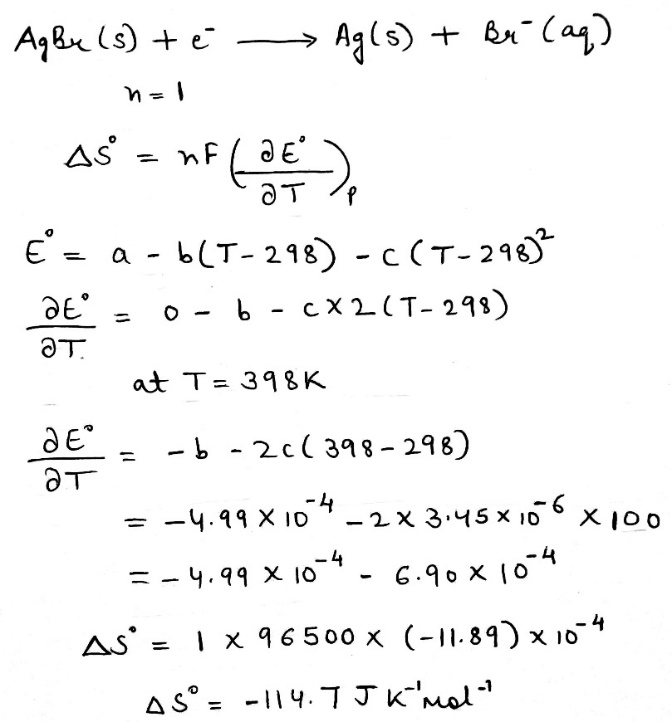
The temperature dependent standard electrode potential of
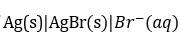
fits the expression




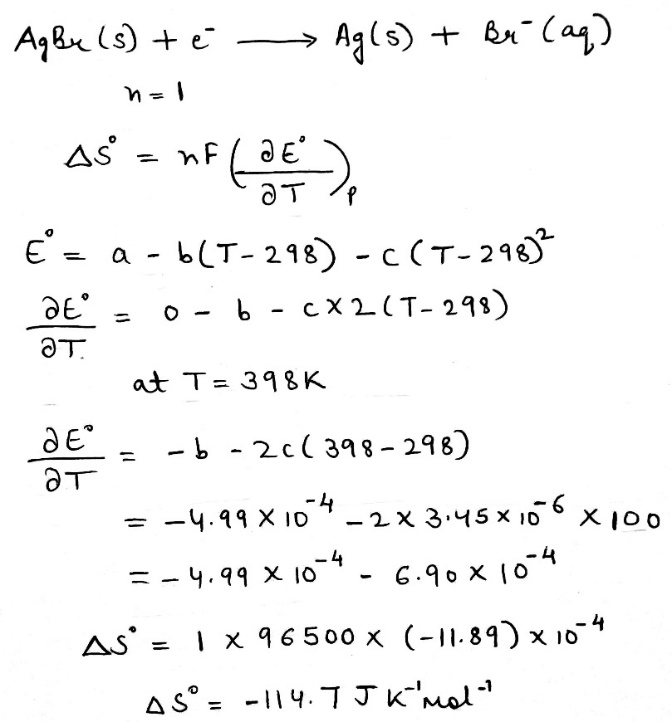
Molar conductivities of an electrolyte are 100 and 50 mScm2 mol-1when concentrations are 4 and 9mM, respectively. The limiting molar conductivity of this electrolyte in mScm2 mol-1is closest to
The E°(M+/M) of the cell, SHE||MX|M can be obtained from the plot of (Ecell is the cell potential and m is the molality of ideal dilute solution of MX)
The amount of

(molecular weight 261.32 amu) required to be added to 500g of a 0.11

solution of

in order to raise its ionic strength to 1.00 is approximately:
For the cell
For a weak electrolyte such as acetic acid, the relation among conductance λ, equilibrium constant (K) and concentration (C) can be expressed as: (λ0is the conductance at infinite dilution)
Consider following terms. Identify those which are relevant to d.c. polarography
A. Thermal current
B. Supporting electrolyte
C. Depolarization
D. Gelatin
Correct answer is
What is the cell potential (in V) at 298 K and 1 bar for the following cell?
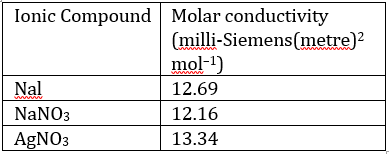
The limiting molar conductivities, at 25°C, of few ionic compounds are given in the table below. The limiting molar conductivity of AgI, in units of milli-Siemens at 25°C is
Given that at 298.15K, At this temperature, the value of is

