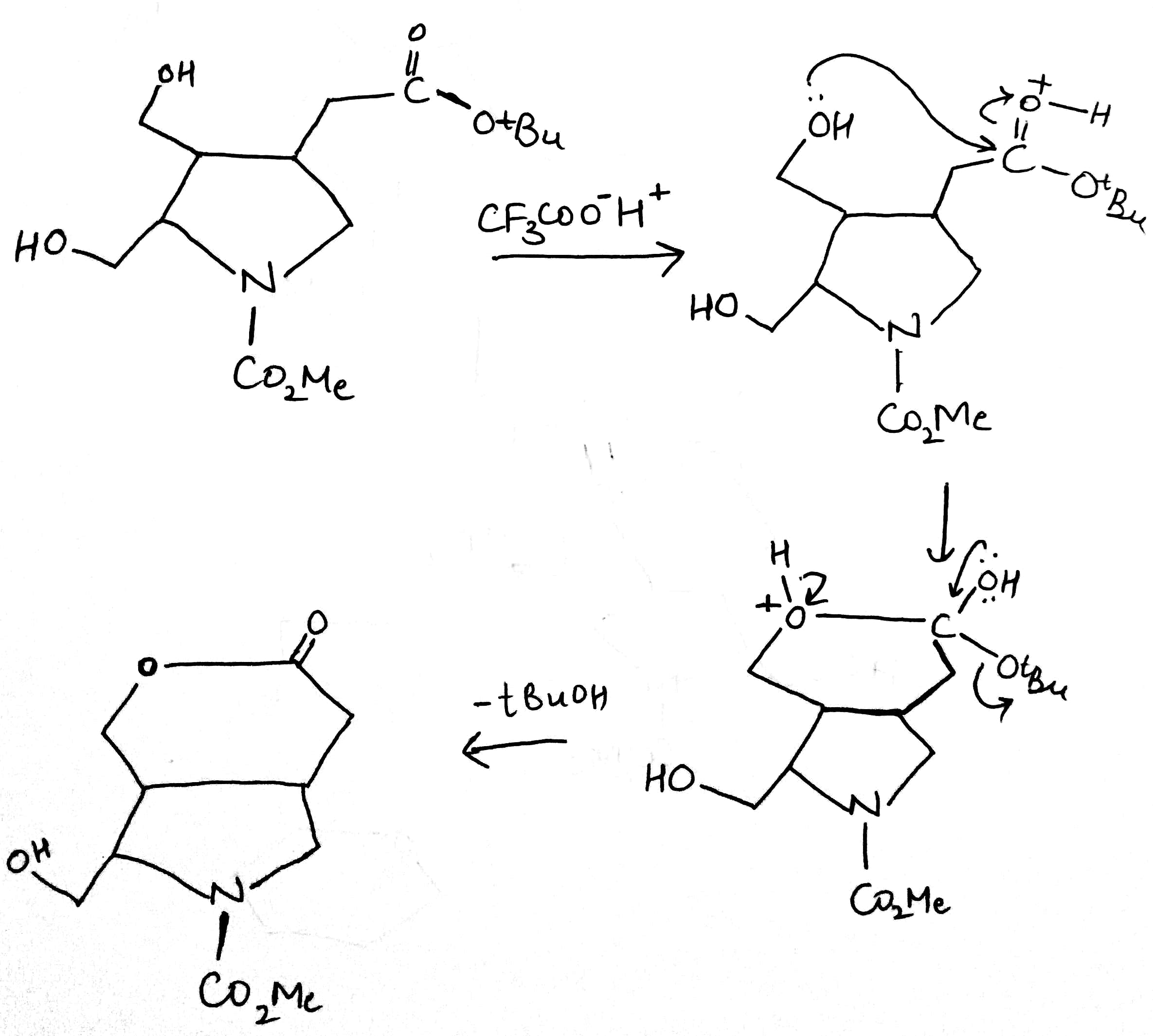
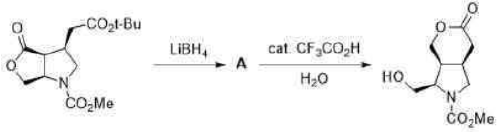
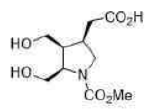
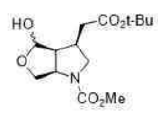
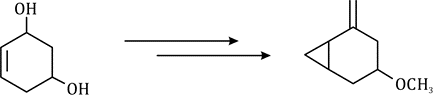
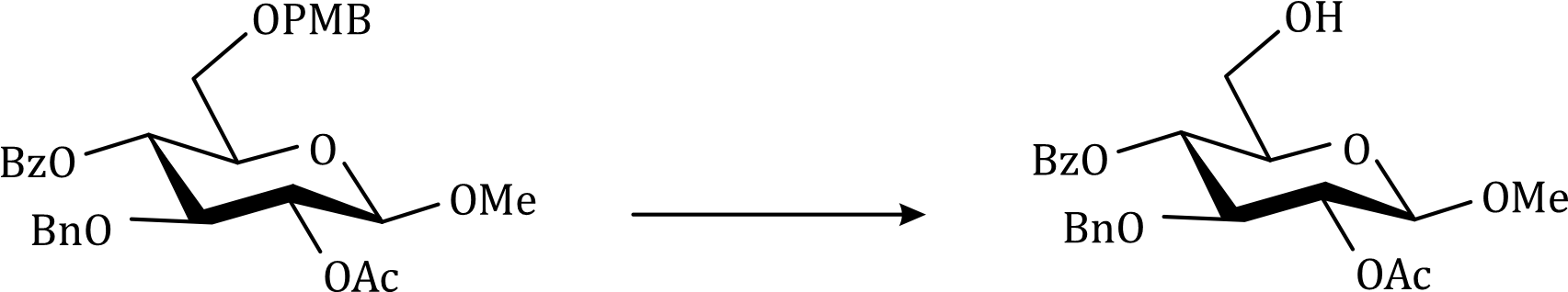
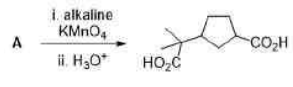
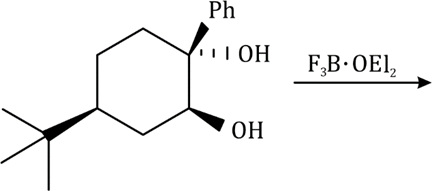
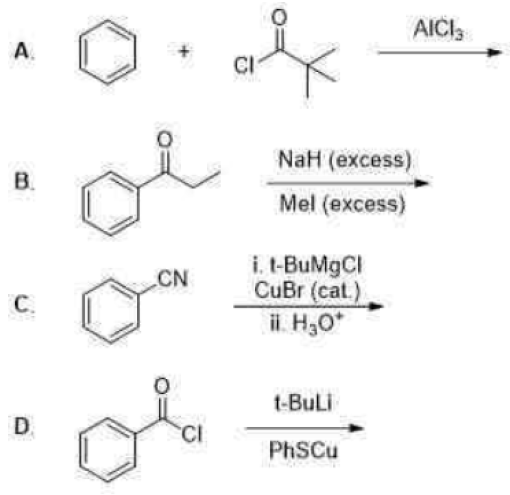
Correct option is D
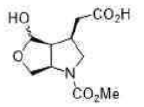
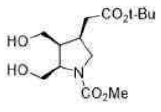
Lithium borohydride (LiBH4) is a borohydride and known in organic synthesis as a reducing agent for esters. The enhanced reactivity in many of these cases is attributed to the polarization of the carbonyl substrate by complexation to the lithium cation. Japanese chemists used an enzyme to make a single enantiomer of the acid below, and were able to reduce either the ester or the carboxylic acid by choosing lithium borohydride or borane as their reagent.
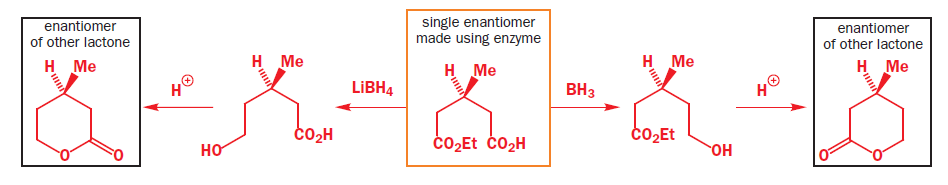
Trifluoroacetic acid (TFA) is a synthetic organofluorine compound with the chemical formula CF3CO2H.Trifluoroacetic acid is a strong acid.