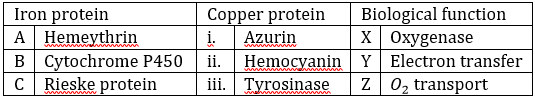
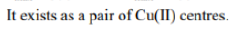Correct option is C
Blue Copper Proteins
A Type 1 centre is characterized by an intense absorption in the electronic spectrum with
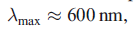
, and
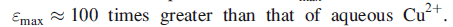
The absorption is assigned to charge transfer from a cysteine ligand to Cu2+. In the EPR spectrum (Cu2+has one unpaired electron), narrow hyperfine splitting is observed.
Blue copper proteins contain a minimum of one Type 1 Cu centre, and those in this class include plastocyanins and azurins.
Single-crystal structural data have provided valuable information about blue copper proteins containing Type 1 Cu centres. Figure shows a representation of the folded protein chain of spinach plastocyanin. The Cu(II) centre lies within a pocket in the chain, bound by a Cys, a
Met and two His residues. The S(Met) atom is significantly further away from the Cu(II) centre than is S(Cys).
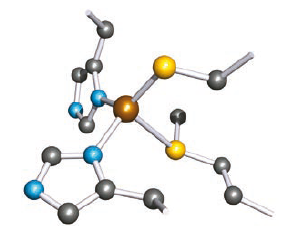
Hydrogen atoms are omitted; colour code: Cu, brown; S, yellow; C, grey; N, blue; O, red.
Figure below shows the backbone of the protein chain in azurin isolated from the bacterium Pseudomonas putida. The coordination environment of the Cu(II) centre resembles that in plastocyanin with Cu-S (Met)>Cu-S(Cys), but in addition, an O atom from an adjacent Gly residue is involved in a weak coordinate interaction.
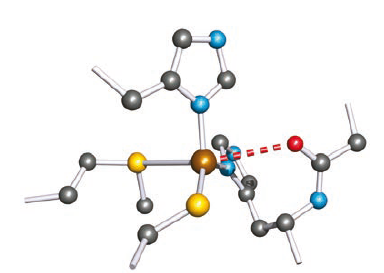
The Cu(II) centre, coordinated by a methionine, a cysteine and two histidine residues; one O atom from the glycine residue adjacent to one of the histidines interacts weakly with the metal centre (the red hashed line).