Correct option is B
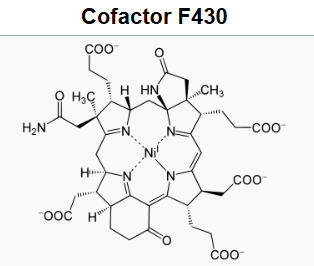
Cofactor F430
F430 is the cofactor (sometimes called the coenzyme) of the enzyme methyl coenzyme M reductase (MCR).
Coenzyme F430 features a reduced porphyrin in a macrocyclic ring system called a corphin. In addition, it possesses two additional rings in comparison to the standard tetrapyrrole (rings A-D), having a γ-lactam ring E and a keto-containing carbocyclic ring F. It is the only natural tetrapyrrole containing nickel, an element rarely found in biological systems.
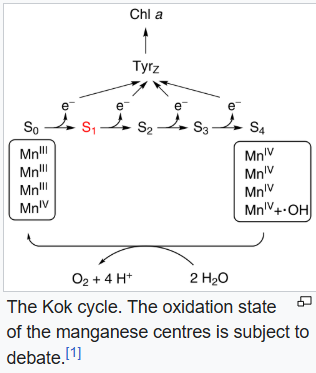
Oxygen-evolving complex
The oxygen-evolving complex (OEC), also known as the water-splitting complex, is a water-oxidizing enzyme involved in the photo-oxidation of water during the light reactions of photosynthesis. OEC is surrounded by 4 core proteins of photosystem II at the membrane-lumen interface. The mechanism for splitting water involves absorption of three photons before the fourth provides sufficient energy for water oxidation. The complex can exist in 5 states, denoted S0 to S4, with S0 the most reduced and S4 the most oxidized. Energy from the photons captured by photosystem II moves the system from state S0 to S1 to S2 to S3 and finally to S4. S4 reacts with water producing free oxygen:
This conversion resets the catalyst to the S0 state. The active site of the OEC consists of a cluster of manganese and calcium. The mechanism of the complex is proposed to involve an Mn-oxide which couples by O-O bond formation to a calcium oxide/hydroxide.
Carboxypeptidase-A
Carboxypeptidase A usually refers to the pancreatic exopeptidase that hydrolyzes peptide bonds of C-terminal residues with aromatic or aliphatic side-chains. Carboxypeptidase A (CPA) contains a zinc (Zn2+) metal center in a tetrahedral geometry with amino acid residues in close proximity around zinc to facilitate catalysis and binding.
Hemerythrin
Hemerythrin is an oligomeric protein responsible for oxygen (O2) transport in the marine invertebrate phyla of sipunculids, priapulids, brachiopods, and in a single annelid worm genus, Magelona. Myohemerythrin is a monomeric O2-binding protein found in the muscles of marine invertebrates. Hemerythrin and myohemerythrin are essentially colorless when deoxygenated, but turn a violet-pink in the oxygenated state. The mechanism of dioxygen binding is unusual. Most O2 carriers operate via formation of dioxygen complexes, but hemerythrin holds the O2 as a hydroperoxide (HO2, or -OOH-). The site that binds O2 consists of a pair of iron centres.