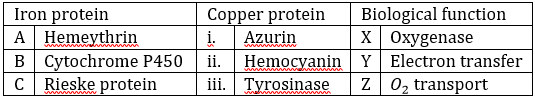
Correct option is D
Oxygenases are enzymes that insert oxygen into other molecules; a monooxygenase inserts one oxygen atom, and a dioxygenase inserts two.
The cytochromes P-450 are metalloenzymes which function as monooxygenases, catalyzing the insertion of oxygen into a C-H bond of an aromatic or aliphatic hydrocarbon, i.e. the conversion of RH to ROH:
Two examples of the biological utilization of this reaction are in drug metabolism and steroid synthesis. The oxygen atom originates from O2: one O atom is inserted into the organic substrate and one atom is reduced to H2O.
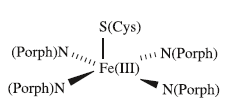
The active site in a cytochrome P-450 is a haem unit. An iron protoporphyrin(IX) complex is covalently bound to the protein through an Fe–S cysteine bond. This has been confirmed crystallographically for cytochrome P-450 complexed with (1S)-camphor . The active site contains a 5-coordinate Fe(III) centre. In its rest state, cytochrome P-450 contains a low-spin Fe(III) centre. Carbon monoxide adducts of cytochromes P-450 absorb at 450 nm and this is the origin of the name of the enzyme. It is proposed that the catalytic cycle for the conversion of RH to ROH follows the sequence of steps:
1.binding of the organic substrate RH to the active site of the metalloenzyme and loss of a bound H2O ligand;
2. 1-electron reduction of low-spin Fe(III) to low-spin Fe(II);
3. binding of O2 to give an adduct, followed by 1-electron transfer from iron to produce an Fe(III)-peroxido complex;
4. acceptance of another electron to give an {Fe(III)-O-O-} species which is protonated to {Fe(III)-O-OH};
5. further protonation and loss of H2O leaving an {Fe(IV)=O} species with the porphyrin ring formally a radical cation;
6. transfer of the oxido O atom to the bound RH substrate and release of ROH with concomitant binding of an H2O ligand to the active site of the metalloenzyme which once again contains low-spin Fe(III).
The insertion of O into the C-H bond of RH is thought to involve a radical pathway.