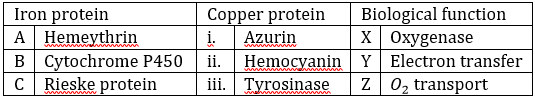
Correct option is A
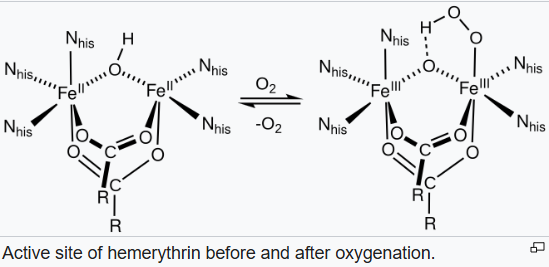
Hemerythrin is a major oxygen-transporting protein in marine invertebrates and has also been proposed to be an oxygen sensor in bacteria. The mechanism of dioxygen binding is unusual. Most O2 carriers operate via formation of dioxygen complexes, but hemerythrin holds the O2 as a hydroperoxide (HO2, or -OOH-). The site that binds O2 consists of a pair of iron centres. The iron atoms are bound to the protein through the carboxylate side chains of a glutamate and aspartates as well as through five histidine residues.
The uptake of O2 by hemerythrin is accompanied by two-electron oxidation of the diferrous centre to produce a hydroperoxide (OOH-) complex. The binding of O2 is roughly described in this diagram:
Hemocyanins are proteins that transport oxygen throughout the bodies of some invertebrate animals. These metalloproteins contain two copper atoms that reversibly bind a single oxygen molecule (O2). Oxygenation causes a color change between the colorless Cu(I) deoxygenated form and the blue Cu(II) oxygenated form.
Cytochromes P450 (P450s or CYPs) are a superfamily of enzymes containing heme as a cofactor that mostly, but not exclusively, function as monooxygenases. P450s are, in general, the terminal oxidase enzymes in electron transfer chains, broadly categorized as P450-containing systems. The term "P450" is derived from the spectrophotometric peak at the wavelength of the absorption maximum of the enzyme (450 nm) when it is in the reduced state and complexed with carbon monoxide.
Tyrosinase is an oxidase that is the rate-limiting enzyme for controlling the production of melanin. The enzyme is mainly involved in two distinct reactions of melanin synthesis otherwise known as the Raper–Mason pathway. Firstly, the hydroxylation of a monophenol and secondly, the conversion of an o-diphenol to the corresponding o-quinone. o-Quinone undergoes several reactions to eventually form melanin. Tyrosinase is a copper-containing enzyme present in plant and animal tissues that catalyzes the production of melanin and other pigments from tyrosine by oxidation.
Rieske proteins are iron–sulfur protein (ISP) components of cytochrome bc1 complexes and cytochrome b6f complexes and are responsible for electron transfer in some biological systems. It is a unique [2Fe-2S] cluster in that one of the two Fe atoms is coordinated by two histidine residues rather than two cysteine residues.
Azurin is a small, periplasmic, bacterial blue copper protein found in Pseudomonas bacteria. Azurin moderates single-electron transfer between enzymes associated with the cytochrome chain by undergoing oxidation-reduction between Cu(I) and Cu(II). Each monomer of an azurin tetramer has a molecular weight of approximately 14kDa, contains a single copper atom, is intensively blue, and has a fluorescence emission band centered at 308 nm.