Correct option is B
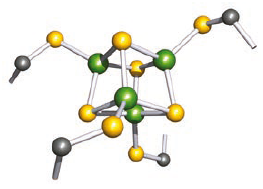
Iron–sulfur proteins are of relatively low molecular weight and contain high-spin Fe(II) or Fe(III) coordinated tetrahedrally by four S-donors. The latter are either S2- (i.e. discrete sulfide ions) or Cys residues attached to the protein backbone. The FeS4 centres occur singly in rubredoxins, but are combined into di-, tri or tetra iron units in ferredoxins. The biological functions of iron–sulfur proteins include electron-transfer processes, nitrogen fixation, catalytic sites in hydrogenases, and oxidation of NADH to [NAD]+ in mitochondria.
Ferredoxins occur in bacteria, plants and animals and are of several types.
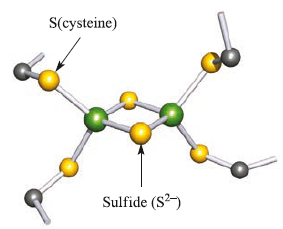
[2Fe–2S] ferredoxins contain two Fe centres, bridged by two S2- ligands with the tetrahedral coordination sphere of each metal completed by two Cys residues.
[3Fe–4S] ferredoxins contain three Fe and four S2- centres arranged in an approximately cubic framework with one corner vacant; this unit is connected to the protein backbone by Cys residues.
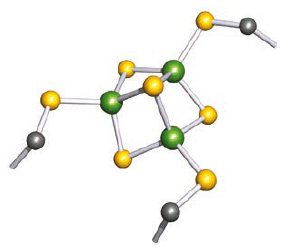
Hydrogen atoms are omitted; colour code: Fe, green; S, yellow; C, grey.
[4Fe–4S] resemble [3Fe–4S] ferredoxins, but contain an additional FeS(Cys) group which completes the approximately cubic cluster core.