Correct option is D
Iron–sulfur proteins are of relatively low molecular weight and contain high-spin Fe(II) or Fe(III) coordinated tetrahedrally by four S-donors. The latter are either S2-(i.e. discrete sulfide ions) or Cys residues attached to the protein backbone. The FeS4centres occur singly in rubredoxins, but are combined into di-, tri or tetra iron units in ferredoxins. The biological functions of iron–sulfur proteins include electron-transfer processes, nitrogen fixation, catalytic sites in hydrogenases, and oxidation of NADH to [NAD]+in mitochondria.
The simplest iron–sulfur proteins are rubredoxins (Mr~6000) which are present in bacteria. Rubredoxins contain single FeS4 centres in which all the S-donors are from Cys residues. The metal site lies in a pocket of the folded protein chain. The four Fe-S(Cys) bonds are of similar length (227–235 pm) and the S-Fe-S bond angles lie in the range 103–1138o. Rubredoxins function as 1-electron transfer sites, with the iron centre shuttling between Fe(II) and Fe(III). Upon oxidation, the Fe-S bond lengths shorten by ~5 pm.
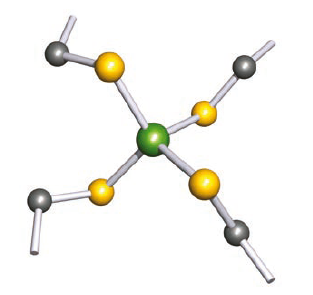
Hydrogen atoms are omitted; colour code: Fe, green; S, yellow; C, grey.
In the reduced state, the oxidation state of Fe is +2. The electronic configuration of Fe2+ is 3d64s0. Hence, Fe2+is paramagnetic.
The distribution of electrons in t2 and e orbitals will be e3t23.
The Jahn–Teller theorem essentially states that any non-linear molecule with a spatially degenerate electronic ground state will undergo a geometrical distortion that removes that degeneracy, because the distortion lowers the overall energy of the species. In octahedral complexes, the Jahn–Teller effect is most pronounced when an odd number of electrons occupy the eg orbitals. The same is true in tetrahedral complexes.