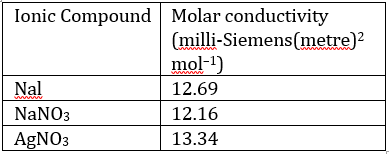
Correct option is C
Fajans' rules, are used to predict whether a chemical bond will be covalent or ionic, and depend on the charge on the cation and the relative sizes of the cation and anion. They can be summarized in the following table:
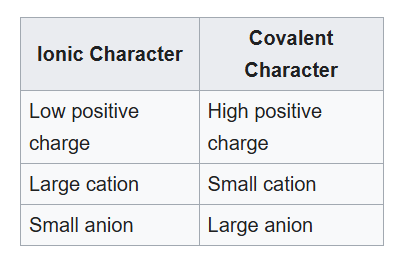
When two oppositely charged ions (X+ and Y- ) approach each other, the cation attracts electrons in the outermost shell of the anion but repels the positively charged nucleus. This results in a distortion, deformation or polarization of the anion. If the degree of polarization is quite small, an ionic bond is formed, while if the degree of polarization is large, a covalent bond results.
If we consider the iodine atom, we see that it is relatively large and thus the outer shell electrons are relatively well shielded from the nuclear charge. In this case, the silver ion's charge will "tug" on the electron cloud of iodine, drawing it closer to itself. As the electron cloud of the iodine nears the silver atom, the negative charge of the electron cloud "cancels" out the positive charge of the silver cation. This produces an ionic bond with covalent character.
When iodine is replaced by fluorine, a relatively small highly electronegative atom. The fluorine's electron cloud is less shielded from the nuclear charge and will thus be less polarizable. Thus, we get an ionic compound (metal bonded to a nonmetal) with a slight covalent character.
Ionic compounds normally dissociate into their constituent ions when they dissolve in water.
Solubility equilibrium is a type of dynamic equilibrium that exists when a chemical compound in the solid state is in chemical equilibrium with a solution of that compound. The solid may dissolve unchanged, with dissociation, or with chemical reaction with another constituent of the solution, such as acid or alkali. Each solubility equilibrium is characterized by a temperature-dependent solubility product which functions like an equilibrium constant.