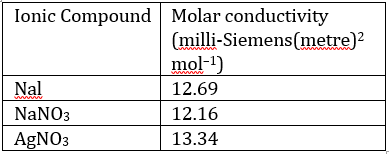
Correct option is B
The electrical resistance of an object is a measure of its opposition to the flow of electric current. Its reciprocal quantity is electrical conductance, measuring the ease with which an electric current passes. Electrical resistance shares some conceptual parallels with mechanical friction. The SI unit of electrical resistance is the ohm (Ω), while electrical conductance is measured in siemens (S) (formerly called the 'mho' and then represented by ℧).
The resistance R of an object is defined as the ratio of voltage V across it to current I through it, while the conductance G is the reciprocal:
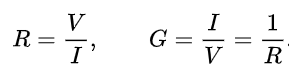
The resistance R and conductance G of a conductor of uniform cross section, therefore, can be computed as
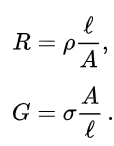
where l is the length of the conductor, measured in metres (m), A is the cross-sectional area of the conductor measured in square metres (m2), σ (sigma) is the electrical conductivity measured in siemens per meter (S·m-1), and ρ (rho) is the electrical resistivity (also called specific electrical resistance) of the material, measured in ohm-metres (Ω·m). The resistivity and conductivity are proportionality constants, and therefore depend only on the material the wire is made of, not the geometry of the wire. Resistivity is a measure of the material's ability to oppose electric current.
The molar conductivity of an electrolyte solution is defined as its conductivity divided by its molar concentration
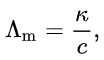
where:
κ is the measured conductivity (formerly known as specific conductance),
c is the molar concentration of the electrolyte.
The SI unit of molar conductivity is siemens metres squared per mole (S m2 mol-1). However, values are often quoted in S cm2 mol-1 . In these last units, the value of Λm may be understood as the conductance of a volume of solution between parallel plate electrodes one centimeter apart and of sufficient area so that the solution contains exactly one mole of electrolyte.