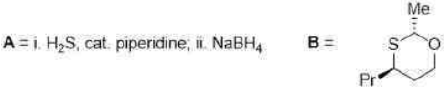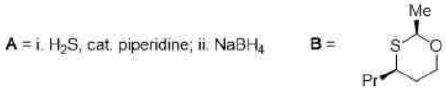Correct option is C
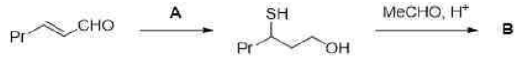
Piperidine is used as a solvent and as a base. Piperidine will abstract H+ from H2S to generate HS- ion. Due to the large size of sulfur, it is highly polarizable. Thus, HS- is soft and will not attack the carbonyl carbon. HS-will attack the softer alkene (conjugate addition or 1,4 addition).
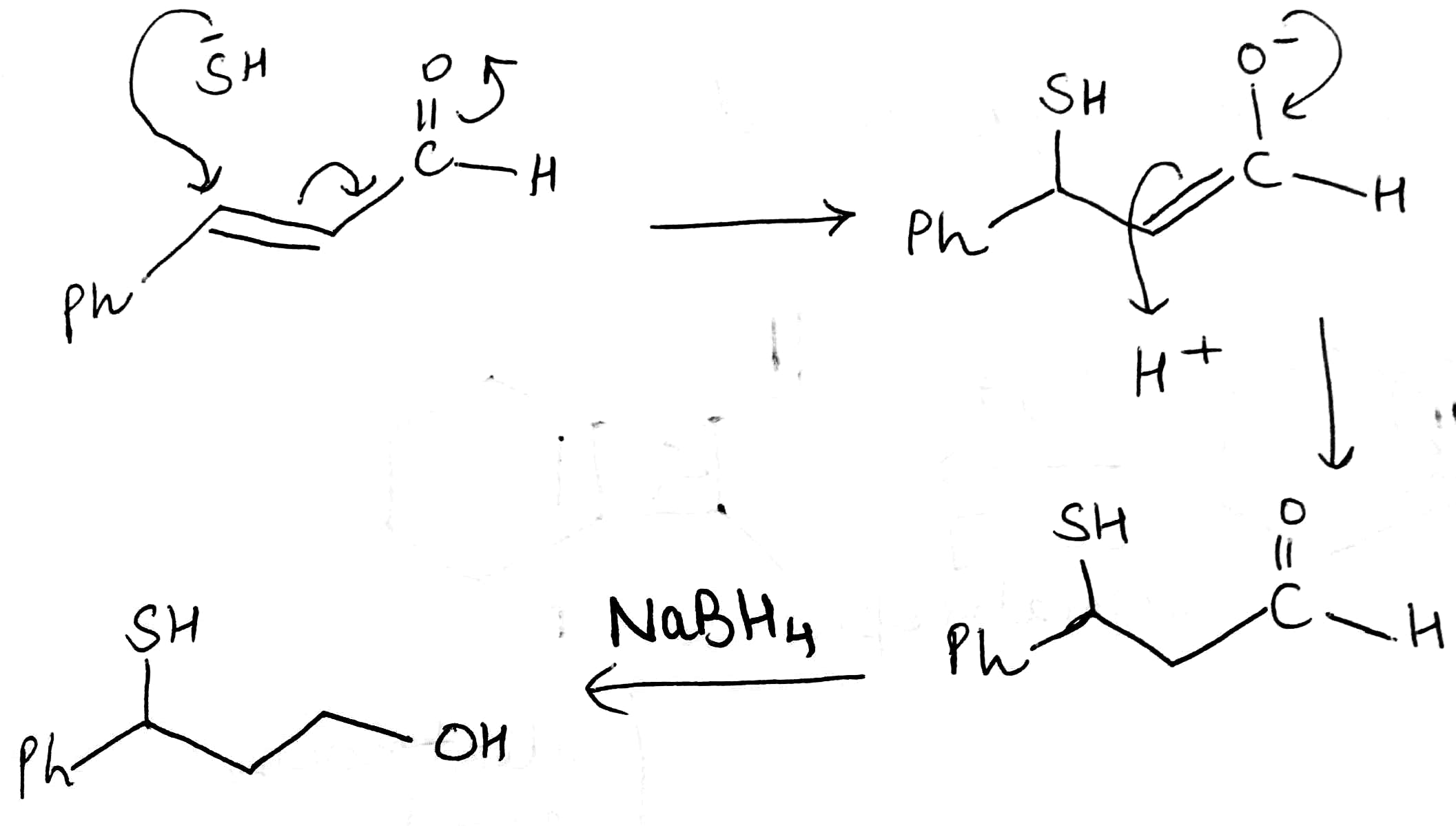
NaBH4 reduces many organic carbonyls, depending on the conditions. Most typically, it is used in the laboratory for converting ketones and aldehydes to alcohols. These reductions proceed in two stages, formation of the alkoxide followed by hydrolysis:
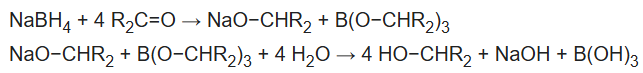
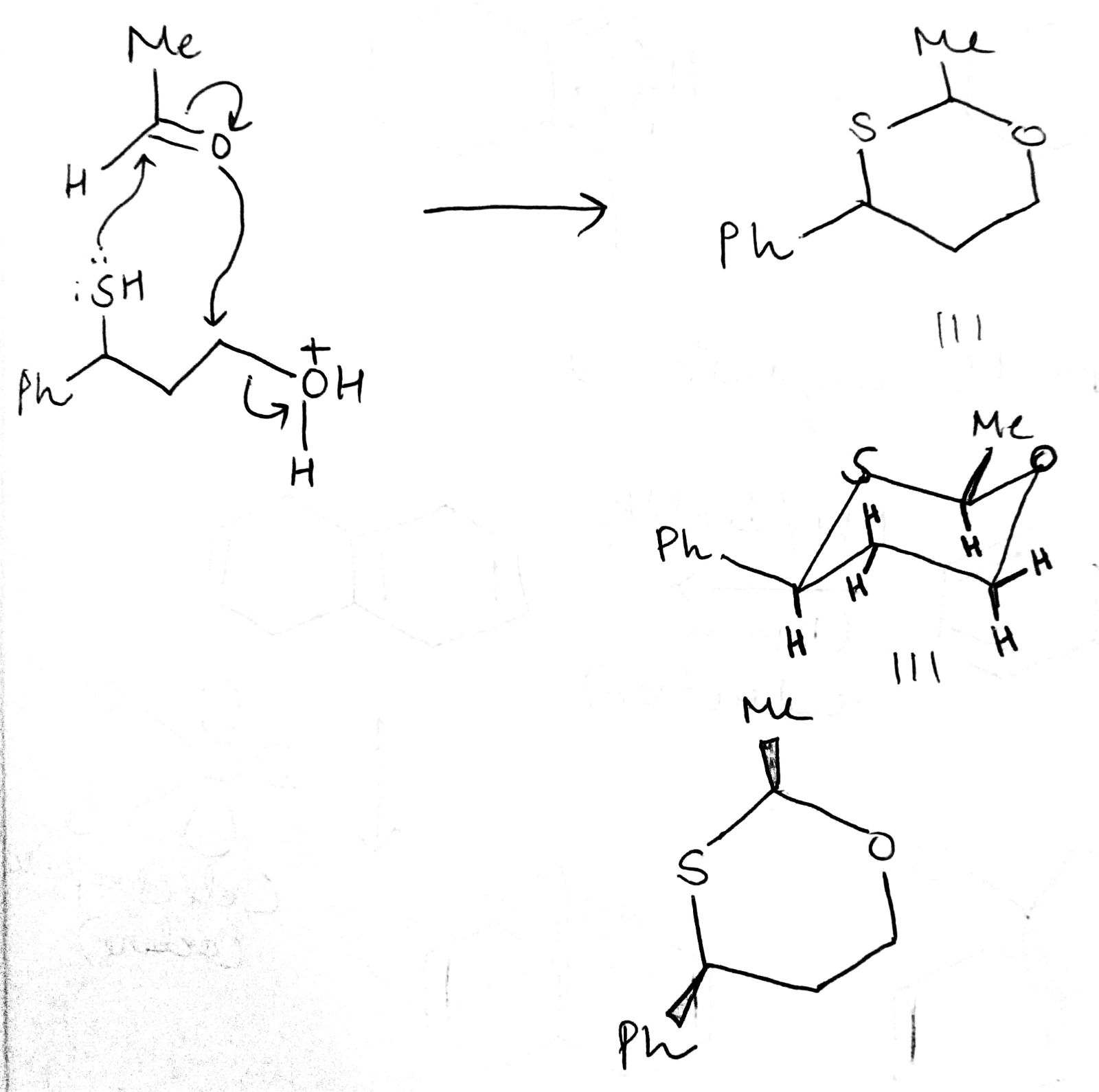
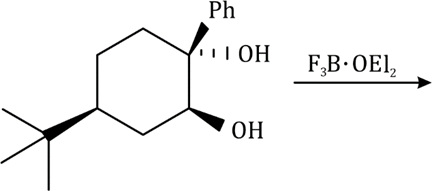
Both Me and Ph are 'up' in the cyclohexane system. Hence, they are cis to each other.
Information Booster
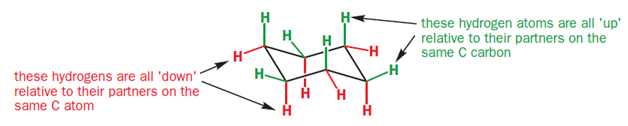
In the chair conformation of cyclohexane, all six carbon atoms are identical, but there are two types of protons-one type stick either vertically up or down and are called axial hydrogen atoms; the other sort stick out sideways and are called equatorial hydrogen atoms.
As you go round the ring, notice that each of the CH2 groups has one hydrogen sticking up and one sticking down. However, all the ‘up’ ones alternate between axial and equatorial, as do all the ‘down’ ones.
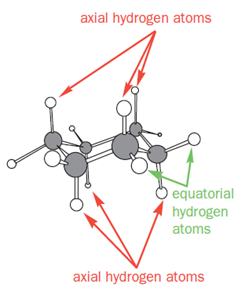
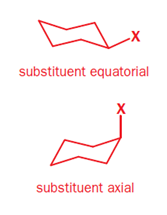
In a monosubstituted cyclohexane, there can exist two different chair conformers: one with the substituent axial, the other with it equatorial. When there are two or more substituents on the ring, stereoisomerism is possible.
Remember, the ‘cis’ and ‘trans’ prefixes merely indicate that both groups are on the same ‘side’ of the cyclohexane ring. Whether the substituents are both axial/equatorial or one axial and the other equatorial depends on the substitution pattern. Each time you meet a molecule, draw the conformation or make a model to find out which bonds are axial and equatorial.
It is not always easy to decide if an equatorial substituent is ‘up’ or ‘down’. The key is to compare it with its axial partner on the same C atom-axial substituents very clearly point ‘up’ or ‘down’. If the axial partner is ‘up’, the equatorial substituent must be ‘down’ and vice versa.