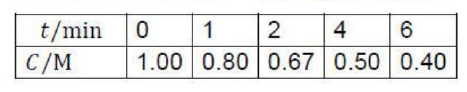
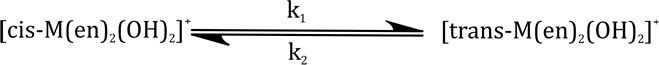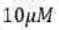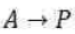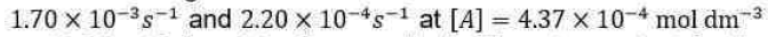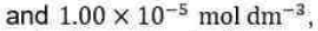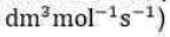Correct option is C
According to this theory, a unimolecular reaction A→P proceeds via the following mechanism:
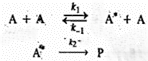
Here A* is the energized A molecule which has acquired sufficient vibrational energy to enable it to isomerize or decompose. In other words, the vibrational energy of A exceeds the threshold energy for the overall reaction A → Products. It must be borne in mind that A* is simply a molecule in a high vibrational energy level and not an activated complex. In the first step, the energized molecule A* is produced by collision with another molecule A. What actually happens is that the kinetic energy of the second molecule is transferred into the vibrational energy of the first. In fact; the second molecule need not be of the same species; it could be a product molecule or a foreign molecule present in the system which, however, does not appear in the overall stoichiometric reaction A → P. The rate constant for the energization step is k1. After the production of A*,it can either be de-energized back to A (in the reverse step, with rate constant k-1) by collision in which case its vibrational energy is transferred to the kinetic energy of an A molecule or be decomposed or isomerized to products (in the step with rate constant k2) in which case the excess vibrational energy is used to break the appropriate chemical bonds.
In the Lindemann mechanism, a time lag exists between the energization of A to A* and the decomposition (of isomerization) of A* to products. During this time lag, A* can be de-energized back to A. According to the steady state approximation (s.s.a), whenever a reactive (i.e., short-lived) species is produced as an intermediate in a chemical reaction, its rate of formation is equal to its rate of decomposition. Here, the energized species A* is short-lived. Its rate of formation=k1[A]2.